Anti-seizure Medication
Specific
drugs
Also see: Anti-seizure Medication Chart
|
Drug |
Dose |
Adverse - neuro |
Adverse other |
Metabolism |
Effect on other meds |
Effect of other meds |
Mechanism |
PBS |
|
Brivaracetam (Briviact) |
Start:
50mg
BD Maintenance: 50-100mg
BD |
Sedation Dizziness Mood
disturbance |
|
CYP2C19
then renal excretion T1/2
– 9hours |
Inc
PHY (20%) |
EI
dec (~20%) |
|
|
|
Cannabidiol (CBD) |
Start: 2.5mg/kg Maintenance: 10-20mg/kg |
Sedation |
Diarrhoea Inc
LFTs |
CYP3A4,
2C19 Inhibits
2C19) T1/2
~60hours Effective
17hrs |
Inc
CLB (~3x) |
|
|
|
|
Carbamazepine (Tegretol)
|
Start:
100-200mg/day Inc
200mg q2/52 Maintenance: 600-1800mg/d QID/BD 15-35mg/kg
child Sz
free (Kwan): 50%
on 400mg 85%
on 600mg 95%
on 800mg |
Sedation (12%) Dizzy/Vertigo (4%) Ataxia
(2%) Headache
(5.5%) Diplopia (1%) Depression
(3.7%) Nausea |
Rash Mild
- 10% Severe/SJS
– predicted by: (HLA)-B*1502
(15% in SE asia) Nausea
2% Wt
gain 2% Aplastic anaemia and
agranulocytosis –
2-5 /million/yr Leukopenia
(usu mild) Thrombocytopaenia Eosinophilia Hepatotoxicity Hyponatraemia |
CYP3A4 Induces
multiple CYP enzymes T1/2
- 25-65hrs then with induction: T1/2
– 12-17hrs |
Dec
VAL Dec
LTG Dec
PHY Dec
OCP |
PHY
dec |
Sodium
channel blocker |
100mg 200mg 100tabs,
2 rpts, Max 200 200mg
CR 400mg
CR 200tabs,
2 rpts 100mg/5ml
liquid, 300ml |
|
Clobazam
(Frisium) |
Start: 5mg bd BD Maintenance: 10-20mg BD |
Sedation |
Rash |
CYP3A4, 2C19 Weak 3A4 inducer T1/2 36-42 |
|
CBD inc |
GABA-A receptor antagonist |
10mg 50tabs (Non-PBS) |
|
Clonazepam (Rivotril) |
Start: 0.25-0.5mg BD Maintenance: Up to 20mg/day |
Sedation |
|
|
|
EI dec |
|
|
|
Ethosuxamide (Zarontin) |
Start: 250mg
BD Maintenance: 20-40mg/kg Max:
1.5g daily |
Worsen GTCS Drowsiness Dizziness Ataxia Headache Euphoria |
Anorexia N+V Epigastric pain Weight loss Hiccup Rash/SJS Blood disorders |
CYP 3A4, 2E1 T1/2 ~60hrs |
|
|
T-type
Ca channels |
250mg 200capsules, 2 rpts (PBS) 250mg/5ml 200ml, 5 rpts, (PBS) |
|
Gabapentin (Neurontin) |
Effective from 900mg daily, slight increase in
efficacy up to 2400mg daily |
Sedation
(12%) Dizziness
(6%) Ataxia (6%) Memory
(2-6%) |
Wt
gain (2-4%) |
RENAL T1/2 – 6hrs |
|
Morphine Inc |
Calcium
channel |
PBS
Code 4928 Refractory partial Sz Capsules 100tabs x5rpts 100mg 300mg 400mg Tablets 100tab x5rpts 600mg 800mg |
|
Drug |
Dose |
Adverse - neuro |
Adverse other |
Metabolism |
Effect on other meds |
Effect of other meds |
|
PBS |
|
Lacosamide (Vimpat) |
Start: 50mg bd Maintenance 100mg – 200mg bd Reduce in renal impairment |
Dizziness (30%) Vertigo, balance disorder Diplopia (7%) No cognitve effects |
Nausea, vomiting and tremor. Mild PR prolongation (CI in heart disease or with type I
anti-arryhtmics) ECG
prior to Rx. |
95% excreted in urine Also: CYP – however is not a major inhibitor or inducer T1/2 – 15hrs |
Nil significnat |
Levels decreased 25% when given with other enzyme
inducers |
Sodium channel - Enhances slow channel inactivation |
Partial sz in combo with 2 other agents (inc. One
second line. Must have failed other
AEDs inc at least one first line and 2 second line agents. 50mg, 100mg, 150mg 14tabs,
1 rpts (titration) - PBS
Code 4271 100mg [56] Rp 5 - PBS 4264 (inital) - PBS 4249
(continuing) 150mg [56] Rp 5 200mg [56] Rp 5 - PBS 4240 (initial) - PBS 4257
(continuing) |
|
Lamotrigine (Lamictal)
|
150-600mg/d 25mg
2/52 50mg
2/52 Then
inc. by 50mg/week With
VPA – half the rate With
inducers – double
the rate. Sz
Free (Kwan) –all types epilepsy: 20%
100mg 85%
200mg 95%
300mg |
Sedation
(8%) Headache
(5%)* Dizziness (4%) Ataxia (4%) Diplopia (1%) Insomnia |
Skin rash (5-10%) SJS(1/500-1/1000) |
|
Nil effect on other meds. OCP – inc. Breakdown 2x LIVER |
VPA decreases metabolism CBZ, OXC, PHY, PHB, PRI Inc. metabolism |
Sodium channel blcoker Enhances rapid inactivation Inhibits Ca2+ channels |
PBS Code 5138 [56]
Rp 5 - for all 5mg 25mg 50mg 100mg 200mg Chewable/dispersable (not scored) |
|
Drug |
Dose |
Adverse - neuro |
Adverse other |
Metabolism |
Effect on other meds |
Effect of other meds |
|
PBS |
|
Levetiracetam (Keppra, Levi) |
Start: 250-500mg bd Seizure freedom by dose (Brodie): 40% 1000mg 54% 2000mg 57% 3000mg |
Sedation (~16%) Aggression (9%) Depression (5%) |
|
|
|
|
SV2A (synaptic vesicle effect) |
PBS code 4928 Refractory partial Sz Tablets
(scored) [60] Rp5 250mg 500mg 1000mg Liquid form available |
|
Oxcarbazepine (Trileptal) |
Start: 300mg bd Seizure freedom by dose: 26% 600mg/d 40% 1200mg/d 50% 2400mg/d |
Dizziness Fatigue Diplopia Somnolence Adverse effects by dose (in excess of placebo): 8% 600mg/d 14% 1200mg/d 22% 2400mg/d |
Rash - Increased risk with HLA B*1502 Hyponatraemia - Severe ~2.5% Vomiting |
Hepatic (No auto-induction like CBZ) T1/2 - 9 |
Dec
VAL Dec
LTG Dec
PHY Dec
OCP |
PHY
dec |
Sodium channel Enhances rapid inactivation Calcium channel block |
PBS code 5183 Partial Sz and primary GTCS not controlled by
others Tablets
Scored [100] Rp 5 150mg 300mg 600mg |
|
Perampanel (Fycompa) |
Start: 2mg/day Maintenance: 4-8mg/day Max: 12mg/day (high dose for use in conjunction with
enzyme inducers) |
On 8mg: Dizziness (26%,P:10%) Somnolence (16%, P6.5%) Fatigue (5.3%, P2.7%) Headache (10%, P 8.6%) Ataxia (6%, P:0%) |
|
Peak 15-120min T1/2 70hours 95% protein bound Steady state takes 2-3 weeks Liver- CYP3A4 Not a major inducer or inhibitor 70% of metabolites excreted in faeces |
|
CBZ, OXC, PHY increase metabolism PHB, PRIM no effect |
AMPA antagonist |
Initial PBS 4656 2mg 7
tabs, 1rpt Focal Sz Must have failed one first line and two second
line agents Must be in combination with two or more AEDs
(inc. One second line) Continuing PBS 4658 All
28 tabs, 5 rpts 4mg 6mg 8mg 10mg 12mg |
|
Phenobarbital |
Start and titrate very slowly e.g. 15mg od, add
15mg every 2/52. Maintenance 60-240mg once daily at night |
|
|
Hepatic and Renal (25-50%) T1/2 -
79hrs Strong CYP 3A4 inducer |
Dec many drugs |
VPA dec |
GABA-A inhibitor |
30mg tablet [200] Rp 4 Scored |
|
Phenytoin (Dilantin)
|
Start 300-400mg/d QID/BD 3-6mg/kg
adult 4-8mg/kg
child |
Dizziness Diplopia Ataxia Incoordination Confusion (cerebellar
degeneration) Worsen
Abs |
Gum
hyperplasia Lymphadenopathy Hirsutism Osteomalacia Facial
coarsening Skin
rash |
CYP
2C9 then 2C19 T1/2
– 22hrs |
Dec
many drugs (EI) |
EI
dec OXC,
TPM Inc |
|
Tablet,
chewable 50mg
[200] Rp 2 Liquid
30mg/5ml 500ml Capsule(Phenytoin
Sodium) 30mg
[200] Rp 2 100mg
[200] Rp 2 |
|
Pregabalin (Lyrica) |
Effective from 150mg daily up to 600mg daily |
|
Weight gain |
Renal excretion T1/2 – 6 hours |
|
|
Calcium channel blocker |
|
|
Primidone |
Start: 100-125mg QID Maintenance: 750-2000mg/day |
|
|
Metabolised to phenobarbital T1/2 – 12 (+79hrs for derived PB) |
|
|
GABA-A |
|
|
|
|
|
|
|
|
|
|
|
|
Drug |
Dose |
Adverse - neuro |
Adverse other |
Metabolism |
Effect on other meds |
Effect of other meds |
|
PBS |
|
Retigabine |
|
|
|
|
|
|
K CNQ +, M-current |
|
|
Rufinamide (Ineolon) |
Start: 200-400mg BD Maintenance: Max 3200mg/day |
|
|
Induced 3A4 |
Dec CBZ Dec LTG Inc PB Inc PHY Inc VPA |
EI Dec |
|
|
|
Sulthiame (Ospolot) |
|
|
|
|
|
|
|
50mg [200], 2rpt, (PBS) 200mg [200], 2rpt, (PBS) |
|
Tiagabine (Gabitril) |
|
|
|
|
|
|
GABA
(+) |
PBS
code 4928 5mg [50], 5rpt 10mg [50], 5rpt 15mg [50], 5rpt |
|
Topiramate
(Topamax) |
Start: 25-50mg
daily Titrate:
25-50mg weekly Maintenance: 100mg
daily (Avg.
dose for control ~175mg) Max: 500mg
daily |
Sedation
(11%) Agitation
etc. (10%) Depression
(8%) Memory
problems (7%) Paraesthesia
(7%+) Confusion
(6%) Behaviour
change (6%) |
Weight
loss (8% of pts) Kidney
stones (1.6%) Acute
Myopia/Glaucoma Decreased
sweating Metabolic
acidosis |
RENAL
(70%) +LIVER |
Dec
OCP (>200mg/day) Inc
PHY (rarely) |
PHY
and CBZ may increase TPX |
Glutamate reduction Na
and Ca |
Treatment of: Seizures 5516 Tablet
25mg, 50mg, 100mg, 200mg [60] Rp 5 Seizures and unable to
take tablets 5173 Capsules
(Sprinkle) 15mg, 25mg, 50mg [60] Rp 5 Migraine 5325 Tablet
25mg, 50mg [60] Rp5 |
|
Valproic acid (Epilim) |
750mg-2g/d BD/QID 20-60mg/kg T1/2
- 7-17hrs Sz
Free (Kwan) –all types epilepsy: 15% 500mg 85%
1000mg 95%
1500mg |
Ataxia Sedation Tremor |
Teratogenicity
Hepatotoxicity
(first 6 months) Thrombocytopenia GIT
irritation Wt
gain Transient
alopecia Hyperamonaemia Pancreatitis
(any time) SJS/TEN/DRESS |
Liver
-
Glucuronidation -P450
Peak
4hrs (7hrs enteric) T1/2
8-20hrs |
Inc
LTG Inc
PHY ?Inc
CBZ |
CBZ
dec PHB
dec |
Inhibits
sodium and calcium channels Enhances
GABA |
100mg
Scored 100tabs,
2 rpts Max
2 packets at once 200mg
Enteric (not scored) 500mg
Enteric (not scored) 100tabs,
2 rpts Max
2 packets at once 200mg/5ml
Liquid |
|
Vigabatrin (Sabril) |
Start: 0.5g/week Maintenance: 2g/day (max 4g) Reduce dose in renal impairment Focal sz only +/- Infantile spasms |
Worsening of generalised epilepsies Visual field defect in 20-40% ** Requires VF’s ** Diplopia Fatigue Sedation Mania Encephalopathy Movement disorders |
|
|
|
|
GABA
+ |
PBS
Code 4929 Sz not controlled by other Rx 500mg [100] Rp 5 Powder for liquid – 60 x 500mg sachets |
|
Zonisamide (Zonegran) Na+, Ca2+ Inc GABA Dec Glutamate |
Monotherapy 100mg od, increase by 100mg every 2 weeks. Adjunctive 25mg bd 1-2 wks 50mg bd 1-2 wks Then inc. By 100mg/wk Maintenance: 300-500mg daily Dec sz freq: 100mg – 18% 300mg – 42% 500mg – 50% |
Dizziness (~10%) Tiredness (<10%) Neuropsychiatric (all types - ~6%) |
Increased Urea and Cr Decreased sweating Weight loss (<7%) Metabolic acidosis – renal stones etc. |
|
Nil |
Inc. Dose may be needed if also taking CBZ, PHY |
|
PBS
4928 Partial Sz, not controlled by others. 25mg [56], 5rpts 50mg [56], 5rpts 100mg [56], 5rpts |
* Probably no
significnat difference from placebo.
Anticonvulsant Identification Chart
|
|
Dose
equivalence (mg) |
IV |
Clearance
t1/2 (Avg) |
Clearance
t1/2 (range) |
|
|
|
|
|
|
Alprazolam |
0.5 |
|
9 |
6-12 |
|
|
|
|
|
|
Clonazepam |
0.5 |
0.5 |
40 |
40 |
|
|
|
|
|
|
Clobazam |
20 |
|
35 |
12-60 |
|
|
|
|
|
|
Diazepam |
10 |
|
60 |
20-100 |
|
|
|
|
|
|
Lorazepam |
1 |
1 |
15 |
10-20 |
|
|
|
|
|
|
Oxazepam |
20 |
|
10 |
4-15 |
|
|
|
|
|
|
Midazolam |
|
2 |
|
|
|
|
|
|
|
|
Temazepam |
20 |
|
15 |
8-22 |
|
|
|
|
|
Mechanism of action
|
Decreased excitation |
Increased inhibition |
|||||
|
Drug/Primary target† |
Na+ channels |
Ca2+ channels |
SV2A |
Glutamate |
GABA |
K+ channels |
|
Carbamazepine |
● |
|||||
|
Phenytoin |
● |
|||||
|
Eslicarbazepine |
● |
|||||
|
Lamotrigine |
● |
|||||
|
Oxcarbazepine |
● |
|||||
|
Rufinamide |
● |
|||||
|
Lacosamide |
● |
|||||
|
Zonisamide |
● |
● |
||||
|
Topiramate |
● |
● |
● |
|||
|
Pregabalin |
● |
|||||
|
Gabapentin |
● |
|||||
|
Vigabatrin |
● |
|||||
|
Benzodiazepines |
● |
|||||
|
Phenobarbital |
● |
● |
● |
|||
|
Valproate |
? |
? |
? |
|||
|
Levetiracetam |
● |
|||||
|
Retigabine |
● |
|||||
|
Perampanel |
● |
|||||
· From AED lecture by Kwan
Drug Interactions/Pharmacokinetics/dynamics
|
ASM |
Elimination |
Action
on cytochromes |
|
Phenytoin |
Hepatic |
Inducer |
|
Carbamazepine |
Hepatic |
Inducer |
|
Valproate |
Hepatic |
Inhibitor |
|
Oxcarbamazepine |
Hepatic (+renal) |
Weak - mixed |
|
Zonisamide |
Hepatic (+renal) |
Nil (Weak –mixed) |
|
Lacosamide |
Hepatic (+renal) |
Weak - mixed |
|
Topiramate |
Renal (+hepatic) |
Weak – mixed |
|
Leviteracetam |
Renal (+hepatic) |
Nil |
|
Gabapentin |
Renal |
Nil |
|
Pregabalin |
Renal |
Nil |
|
Lamotrigine |
Hepatic |
Nil |
|
Perampanel |
Hepatic |
Nil |
|
|
Induces CYP |
Inhibits CYP |
Metabolized
by CYP |
Induces UGT |
Inhibits UGT |
Metabolised
by UGT |
|
Carbamazepine |
2C9, 3A |
No |
1A2, 2C8, 2C9, 3A4 |
Yes |
No |
No |
|
Gabapentin |
No |
No |
No |
No |
No |
No |
|
Lamotrigine |
No |
No |
No |
Yes |
No |
Yes |
|
Levetiracetam |
No |
No |
No |
No |
No |
No |
|
Oxcarbazepine |
3A4/5 |
2C19 |
No |
Yes |
Weak |
Yes |
|
Phenobarb |
2C, 3A |
Yes |
2C9, 2C19 |
Yes |
No |
No |
|
Phenytoin |
2C, 3A |
Yes |
2C9, 2C19 |
Yes |
No |
No |
|
Perampanel |
No |
No |
3A4 |
?No |
?No |
?No |
|
Tomiramate |
No |
2C19 (mild) |
No |
No |
No |
|
|
Valproate |
No |
2C9 |
2C9, 2C19 |
No |
Yes |
Yes |
|
Zonisamide |
No |
No |
3A4 |
No |
No |
Yes |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
T ½ |
Peak |
|
Carbamazepine |
5-26h |
4-8h |
|
Levetiracetam |
6-8h |
0.5-2h |
|
Lamotrigine |
12-60 |
1-3h |
|
Topiramate |
19-25h |
2-4h |
|
Zonisamide |
50-70h |
2.4-4.6h |
COST
|
|
Pack |
Cost/day Maintenance |
Cost/year |
|
|
|
|
|
|
Leviteracetam |
1g- 60- $39.71 |
$1.47 (2g) |
$ 536.55 |
|
Lamotrigine |
100mg- 56 - $27.53 |
$0.98 (200mg) |
$ 357.7 |
|
Carbamazepine |
200mg – 200 -$30 |
$0.30 (400mg CR) |
$ 109.50 |
|
Zonisamide |
100mg -112 - $75.22 |
$2.69 (400mg) |
$ 981.85 |
|
Perampanel |
8mg – 28 - $188.80 |
$6.74 (8mg) |
$ 2460.10 |
|
Brivaracetam |
25mg – 56 – 163.75 |
$5.84 (50mg/day) |
$ 2131.6 |
|
Lacosamide |
200mg – 56 $302.25 |
$10.79 (400mg) |
$ 3938.35 |
Efficacy summary
SANAD at 4 years:
|
|
Adverse effect failure % |
Inadequate control % |
Still on drug % |
|||
|
|
Focal |
Gen |
Focal |
Gen |
Focal |
Gen |
|
CBZ |
31 |
|
18 |
|
50 |
|
|
GAB |
20 |
|
39 |
|
39 |
|
|
LTG |
20 |
14 |
24 |
16 |
54 |
50 |
|
TPX |
31 |
29 |
25 |
21 |
42 |
49 |
|
OXC |
26 |
|
24 |
|
49 |
|
|
VPA |
|
21 |
|
31 |
|
60 |
|
|
|
|
|
|
|
|
Contraception
|
Decrease contraception |
|
|
Phenytoin Carbamazepine Oxcarbamazepine Barbituates Phenobarbitone Primidone Eslicarbazepine Felbamate |
Induce CYP
3A4 enzymes – reduce oestrogen and progestins |
|
Some effect on contraception |
|
|
Topiramate |
Minor
decrease if dose >200mg |
|
Lamotrigine |
Decrease
progestins |
|
Perampanel |
Decreased
progestins at high dose (12mg only, caution over 8mg) |
|
No effect on contraception |
|
|
Valproate Conazepam Levetiracetam
Gabapentin Pregabalin Vigabatrin Ethosuxamide Lacosamide Zonisamide |
|
|
Affected by OCP |
|
|
Lamotrigine |
OCP (containing oestrogens) lowers lamotrigine
level by 40-60% Level can rise during pill free week. Not affected by progeserone. |
|
Method |
Interaction? |
Solution |
|
Combined OCP |
Variable
(see above) |
Try higher
strength oestrogen (e.g. 50mcg) – risk of failure still higher – 3/1000yrs vs
0.3/1000 yrs Taking active
pills with a break every 3 months also likely to be more effective. |
|
Progesterone
only pill |
Variable
(see above) |
Double dose
at least. Effectiveness still may be
less than usual. |
|
Morning after pill |
|
Double first
dose and give a single second dose 12hours later |
|
Implanon |
Yes –
decreased effectiveness |
Not-safe |
|
Depo-provera |
None |
Safe (some
reccomend dosing every 10 weeks rather than 12 weeks for extra safety) |
|
IUDs |
None |
Safe |
Pregnancy
Effect of medication on pregnancy
Risk of Major Congenital Malformation (MCM) if exposed to monotherapy during pregnancy
|
|
EURAP 2024 |
|
N Am. AED reg. |
UK/ Ireland Reg 2014 |
Aust. Reg 2019 |
AAN 2024 Combined |
Other data |
|||||
|
|
Dose mg/day |
MCM % |
MCM % |
Dose mg/day |
MCM % |
MCM % |
|
|
|
|||
|
Baseline risk |
|
?3.5 |
1 (control) |
|
|
3.7 |
|
|
|
|||
|
|
|
|
|
|
|
2.8 |
|
|
|
|||
|
Valproate |
Overall |
9.9 |
9.3 |
Overall |
6.7 |
14.8 |
9.67 |
|
||||
|
</=650 |
6.0 |
</=600 |
5.0 |
|
|
|||||||
|
650-1450 |
11.1 |
>600-1000 |
6.1 |
|
|
|||||||
|
>/=1450 |
25.2 |
>1001 |
10.4 |
|
|
|||||||
|
Carbamazepine |
Overall |
5.4 |
3 |
Overall |
2.6 |
5.9 |
4.37 |
|
|
|||
|
</=700 |
4.6 |
</=500 |
1.9 |
|
|
|||||||
|
>700-1000 |
5.9 |
>500-1000 |
2.7 |
|
|
|||||||
|
>1000 |
9.1 |
>1001 |
5.3 |
|
|
|||||||
|
Lamotrigine |
Overall |
3.1 |
1.9 |
Overall |
2.3 |
4.9 |
3.07 |
|
|
|||
|
</=325 |
2.5** |
</=200 |
2.1 |
|
|
|||||||
|
>325 |
4.3** |
>200-400 |
2.4 |
|
|
|||||||
|
|
|
>401 |
3.4 |
|
|
|||||||
|
Phenytoin |
|
6.3 |
2.9 |
|
3.7 |
2.3 |
5.13 |
|
|
|||
|
Levetiracetam |
|
2.5 |
2.4 |
|
0.7 |
3.6 |
3.48 |
|
||||
|
Topiramate |
|
4.9 |
4.2 |
|
4.3 |
1.9 (14.1 in
polytherapy) |
4.45 |
|
||||
|
Gabapentin |
|
|
0.7 |
|
|
|
3.09 |
|
|
|||
|
Oxcarbazepine |
|
2.9 |
2.2 |
|
|
5.3 |
3.13 |
|
|
|||
|
Phenobarbital |
Overall |
6.2 |
5.5 |
|
|
|
6.03 |
|
|
|||
|
</=60 |
2.6 |
|
|
|||||||||
|
60-130 |
6.1 |
|
|
|||||||||
|
>130 |
10.8 |
|
|
|
||||||||
|
Zonisamide |
|
3.4 |
|
|
|
|
3.92 |
McCluskey et al Seizure 2021 UK database -
Monotherapy MCM13% (3 cases) -
Polytherapy MCM 6.9% -
21% small for gestational age |
|
|||
|
Perampanel |
|
? |
|
|
|
|
? |
Vazquez 2020 AAN poster -
99 pregnancies (EURAP) -
45 reached term – no MCM |
|
|||
|
Lacosamide |
|
0 (NS) |
|
|
|
|
|
|
|
|||
|
Ethosuxamide |
|
0 (NS) |
|
|
|
|
? |
|
|
|||
|
Clobazam |
|
|
|
|
|
|
3.13 |
|
|
|||
|
Clonazepam |
|
|
|
|
|
|
3.03 |
|
|
|||
* Hernandez-diaz Annals of
neurology 2017
** 2018 data
AAN Data
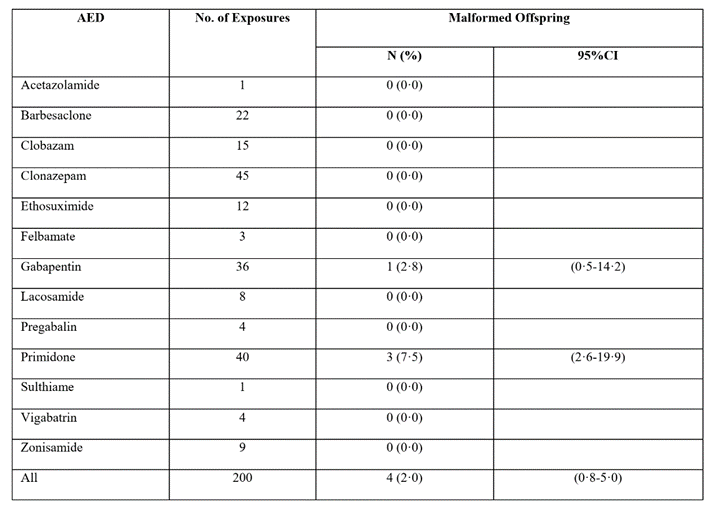

Risk of developmental disorders and other outcomes if exposed to monotherapy during pregnancy
|
|
NEAD |
Liverpool MNDG 2015 |
Small for gestational age* NEAD |
Other data |
|
|||
|
|
IQ at 4.5yrs |
IQ at 6yrs |
IQ at 6 years |
Risk ratio (adjusted) |
% |
|
|
|
|
Baseline risk |
|
|
Controls 107 |
|
5 |
|
|
|
|
|
|
|
No AED 104 |
|
|
|
|
|
|
Valproate |
96* |
98* <1g-104 >1g-94 |
</=800mg - 98 |
1.5 |
11 |
Associated with increased risk of autism and ADHD Decreased cortical thickness Significant increased risk of neural tube defects |
||
|
>800mg - 93 |
||||||||
|
Carbamazepine |
106 |
105 |
105 |
1.25 |
9 |
|
||
|
Lamotrigine |
106 |
108 |
103 |
1 |
8 |
|
||
|
Phenytoin |
105 |
108 |
|
0.8 |
5 |
|
|
|
|
Levetiracetam |
|
|
|
1.25 |
9 |
Study of 51 patients – similar developmental outcomes to controls |
|
|
|
Topiramate |
|
|
|
2.4 |
19 |
Quite high risk of small birth size Australian registry suggested particularly high risk if topiramate is
used in polytherapy. |
|
|
|
Gabapentin |
|
|
|
1.2 |
9 |
|
|
|
|
Oxcarbazepine |
|
|
|
1.4 |
11 |
|
|
|
|
Phenobarbital |
|
|
|
2.4 |
17 |
|
|
|
|
|
|
|||||||
|
|
|
|||||||
|
|
|
|||||||
|
Zonisamide |
|
|
|
1.9 |
15 |
McCluskey et al Seizure 2021 UK database -
Monotherapy MCM13% (3 cases) -
Polytherapy MCM 6.9% -
21% small for gestational age |
|
|
|
Perampanel |
|
|
|
|
|
Vazquez 2020 AAN poster -
99 pregnancies (EURAP) -
45 reached term – no MCM |
|
|
|
Lacosamide |
|
|
|
|
|
|
|
|
|
Ethosuxamide |
|
|
|
|
|
|
|
|
|
Clobazam |
|
|
|
|
|
|
|
|
|
Clonazepam |
|
|
|
|
|
|
|
|

Other factors affecting risk
• If a women has had a previous child with malformation on a certain drug then risk of recurrent malformation is much higher
o This has been demonstrated for valproate in Australian data – e.g. risk at 1000mg/day goes from ~11% to 27%
Type of malformations
From UK registry JNNP 2014
Folate
· NEAD 6 yr study showed
improved IQ for periconceptional folate use (mean IQ - 108 vs 101)
· Folate
supplementation from 4 weeks pre-conception and during first trimester
associated with OR 3.9 reduction in risk of language delay (MoBa Study - Husbye
et al Neurology 91:2018)
· Rare evidence that
over-supplementation may adversely affect neurodevelopment (Murray et al. Nutr
Rev 2017)
·
Dose
o
Literature supports 0.4mg-4mg/day
o
Proposed (RBWH) 1mg/day for 3 months prior to pregnancy
and 0.5mg/day through-out pregnancy
Pregnancy Studies
EURAP study
· European
· Published updates
2011, 2018, 2014
NEAD study
· Pregnant women on
monotherapy 199-2004
· F/U of child
development
· ~300 patients
· 4.5year F/U (Neurology 2012, 78:1207)
· 6 year F/U (Lancet
Neurology 2013 12:244)
North American AED registry
· Neurology
2012;78:1692–1699
UK Ireland AED registry Levetiracetam results
· Neurology
2013;80:400–405
UK EPR subset
• Neurology 2014: 82:213
• Developmental scales - control vs LEV vs VPA
Effect of pregnancy on medication
|
|
Reduction in serum concentration |
Reduction in serum concentration (MONEAD study) JAMA Neurol 2022 |
|
|
Carbamazepine |
?unchanged |
Minimal change |
|
|
Lamotrigine |
50% (17%-70%) |
56% |
Increase in oestrogen driven glucuronidation Returns to normal within a few days post-partum MONEAD study found significant decrease in first trimester |
|
Levetiracetam |
50% (40-60%) |
36.8% |
Increased GFR and renal clearance Increased from first trimester then stable (MONEAD study found significant decrease in concentration from first trimester) |
|
Lacosamide |
|
40% |
|
|
Phenytoin |
40% |
N/A |
|
|
Phenobarbital |
40% |
N/A |
|
|
Oxcarbazepine |
35% |
No change
(unbound) |
|
|
Topiramate |
35% |
Reduction NS |
|
|
Zonisamide |
30% |
29.8% |
|
|
Perampanel |
?2-4x decrease |
|
|
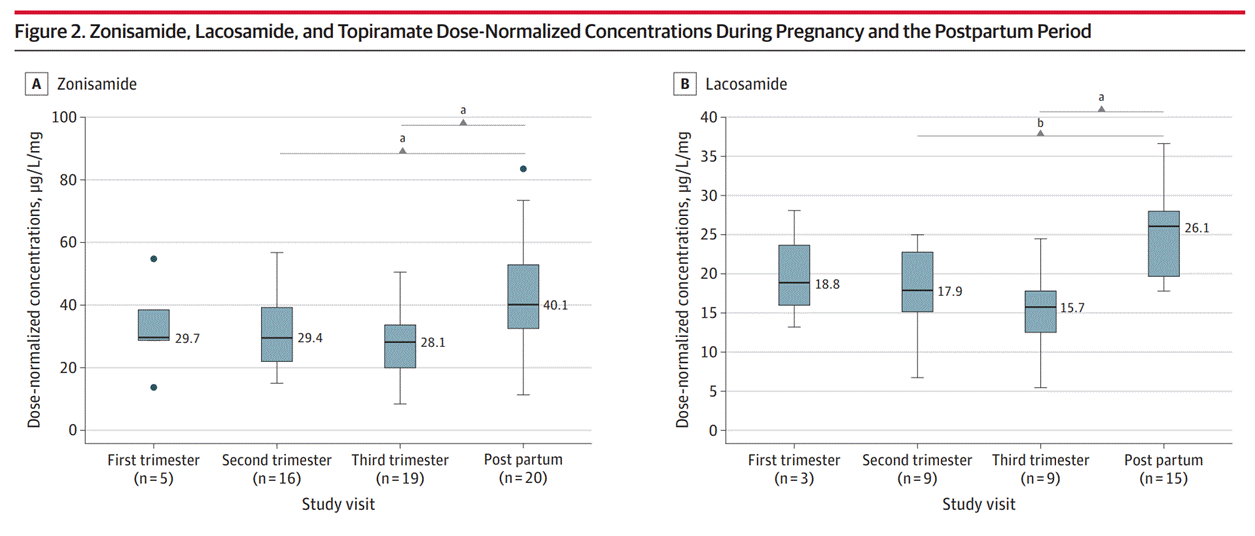
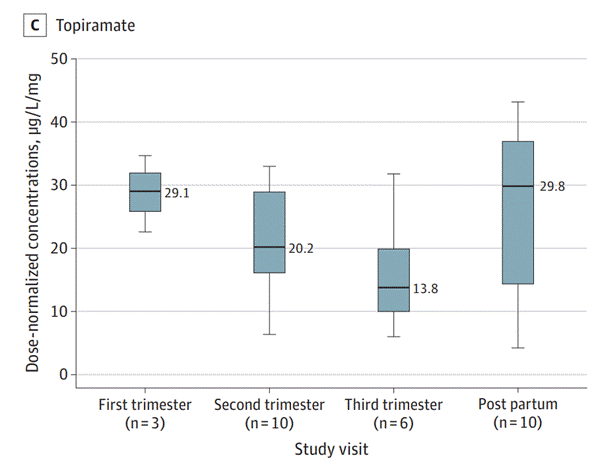
Lamotrigine
• Increase in oestrogen driven glucuronidation
• Decreases by a variable 60% during pregnancy
• Returns to normal within a few days post-partum
o Aim to return to long term dose by day 10
o Consider slightly higher dose (~extra 50mg) compared to pre-pregnancy
MONEAD Study results

Breast Feeding
• Overall data suggests IQ of breastfed infants is higher than non-breast fed – even if the mother is taking AED
|
Drug |
Dose (%)
acquired from breast milk ** |
Infant to mother
concentration*** |
|
|
Phenobarbital |
>50 |
|
|
|
Lamotrigine |
>50 |
30% (up to ~80%) |
50 infants – one apnoea with very high dose, IQ studied in 30 children
– no effect |
|
Levetiracetam |
High transfer to
milk, but low infant serum conc. |
5% (up to 20%) |
No adverse effects noted in small numbers. |
|
Ethosuximide |
>50 |
|
|
|
Topiramate |
High transfer to
milk, but low infant serum conc. |
|
No adverse effects noted in small numbers. |
|
Carbamazepine |
<5 |
5% (up to 6%) |
|
|
Phenytoin |
<5 |
|
|
|
Valproate |
<5 |
|
|
|
Zonisamide |
High milk
transfer, high infant dose |
45% (?up to 65%) |
No adverse effects noted in small numbers. |
|
Gabapentin |
High milk
transfer, low infant serum conc. |
|
6 infants – no adverse effects |
** This is the % of the minimum recommended infant dose that a fully
breast-fed infant would acquire.
*** MONEAD study (Birnbaum JAMA Neurol 2019)
• Phenobarbitone is the only medication which is clearly likely to be a problem and can cause drowsiness etc. in the infant.
Specific drugs
Brivaracetam
Mechanism
• Binding to SV2A – 20x affinity compared to levetiracetam
• Higher brain permeability than levetiracetam
•
Metabolism
• T1/2 – 9 hours
• Renal excretion after extensive metabolism in liver – hydrolysis and to a lesser extent CPY2C19
Interactions
• Clearance increase by enzyme inducers
• May increase carbamazepine and phenytoin levels by up to 20%
• Overall
no clinically significant interaction with any other AEDs noted
• No
effect on oral contraceptive pill
• Not effective in combination with levetiracetam
Side effects
|
|
Diff from placebo % |
BRIV |
Placebo |
|
Somnolence |
6.7 |
15.2 |
8.5 |
|
Dizziness |
4.0 |
11.2 |
7.2 |
|
Fatigue |
5.0 |
8.7 |
3.7 |
|
Irritability |
2.1 |
3.2 |
1.1 |
Efficacy
• Pooled analysis of Phase 3 trials (as an adjunctive therapy for focal epilepsy) (2016):
|
|
Placebo |
50mg/day |
100mg/day |
200mg/day |
|
% reduction in Sz
over placebo |
- |
19.5 |
24.4 |
24.0 |
|
>50% reduction |
20.3 |
34.2 |
39.5 |
37.8 |
|
Seizure free |
0.5 |
2.5 |
5.1 |
4.0 |
• Efficacy from pooled phase 3 trials as adjunctive treatment for focal epilepsy (2016):
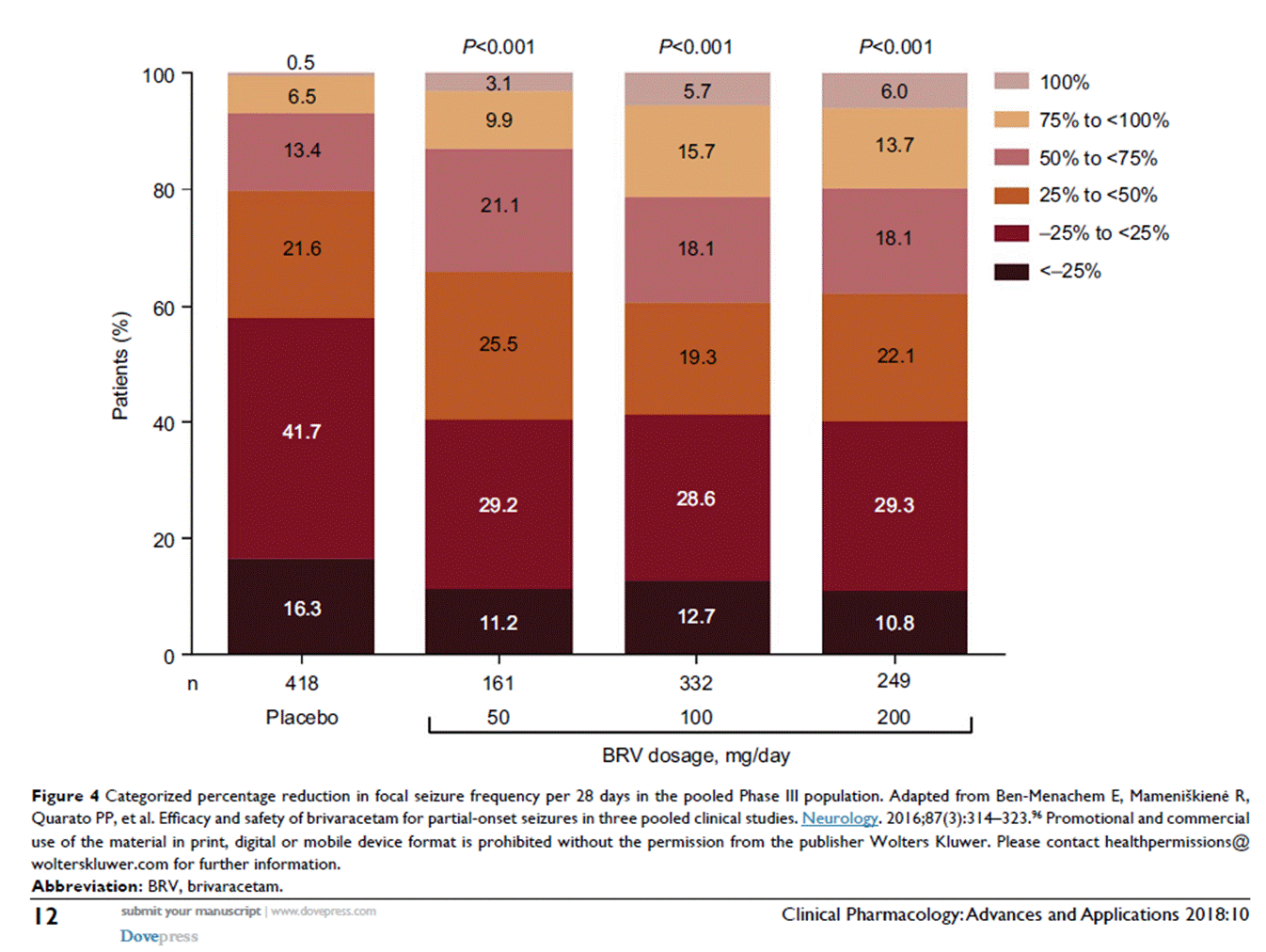
• Response over time from pooled phase 3 trials as adjunctive treatment for focal epilepsy (2016):
o Conclusion: ?higher dose achieves faster response, however in the end no difference

Dosing
• Statistically significant difference in response from 50mg/day vs 100mg/day, however no overall benefit in going to 200mg/day
• Side effects slightly higher on 100mg/day vs 50mg (however no great difference between 100mg/day and 200mg/day)
• No demonstrated need for titration
• Based on above data would suggest
o Start 50mg/day and increase to 100mg/day if no response after 1-2 months
o Should rarely consider going to 200mg/day, however unlikely to cause worsening of side effects.
• Comes as 25, 50, 75, 100mg tablets and 10mg/ml liquid
• Cost: $163.75 /month for 50mg/day.
Zonisamide
• Synthesized in 1974
• Licensed in Japan in 1989, USA 2000
• Peak plasma 2-6hrs
• Bioavailability >95% (not affected by food)
Interactions
o Half life reduced by phenytoin, phenobarbitone and carbamazepine (which induce 3A4)
o Does not effect plasma levels of other AEDs
• Elimination – hepatic/renal
o Metabolised by CYP3A4
• Haltf life 63hours
Side effects
• Side effects shown to have a dose dependent effect include:
• Dizziness (12%)
• Somnolence (14%)
• Tiredness (8.5%)
• Weight loss (7%)
• Other reported side effects
• Nausea and vomiting
• Drop out due to side effects ~27% at 500mg dose
• Serious adverse effects
• Metabolic acidosis
• Similar to placebo
Effectiveness
• Trial as add on in CPS (2005)
• ZNS 500 mg/day produced a greater median reduction in CPS frequency than placebo (51.2% vs 16.3%, p<0.0001)
• 300mg and 500mg produced statistically significant effects, 100mg did not
• Trial in mixed population of focal and generalised seizures showed efficacy in both groups (higher in generalised epilepsy group!)
Dosing
• Initially, 25 mg twice daily; increase after 1 week to 50 mg twice daily; then increase, at weekly intervals, by up to 100mg daily according to response.
•
• Maintenance, usually 300–500 mg daily in 1 or 2 doses.
•
• Lower doses may be effective in some people, including those not taking carbamazepine, phenytoin or other CYP3A4 inducers.
•
• CrCl <20 mL/minute, mild-to-moderate hepatic impairment, patients not taking a CYP3A4 inducer, titrate dosage at intervals of 2 weeks.
Lacosamide
Initially, 50 mg twice daily, increased after 1 week to 100 mg twice
daily.
If needed, increase dose by 50 mg twice daily each week; maximum 200 mg
twice daily.
Evidence that combination with non-sodium channel blocking drugs:
increases efficacy (50% seizure reduction response rate with 400mg daily was
40% vs 62%) and decreases side effects.
(Sake et al CNS drugs 2010 post hoc analysis)
Oxcarbazepine
Barcs G, Walker EB, Elger CE, et al. Oxcarbazepine placebo-controlled,
dose-ranging trial in refractory partial epilepsy. Epilepsia.
2000;41(12):1597-1607. doi:10.1111/j.1499-1654.2000.001597.x
Perampenal
Mechanism
Pharmacokinetics
•
|
|
Monotherapy |
CYP3A4 inducer |
|
Absorption |
Tmax 0.5-2.5h |
|
|
Food |
Tmax delayed 2-3hours |
|
|
Bioavailability |
100% |
|
|
Protein binding |
95% |
|
|
Distribution |
Vd =1.1L/kg (body water) |
|
|
Plasma Half life |
48hours |
25hours |
|
Time to steady state |
10-19days |
|
|
Metabolism |
CYP3A4 to various metabolites |
|
|
Route of elimination |
70% faeces, 30% urine |
|
|
Plasma target range |
180-980ug/L (515-2803nmol/L) |
|
|
Interactions |
|
|
|
Inducer |
No |
|
|
Inhibitor |
No |
|
|
Of perampanel on others: |
35% increase in oxcarbazepine, small decrease in OCP at high dose, otherwise minimal impacts |
|
|
Effect of other drugs on peramanel: |
CYP3A4 enzyme inducers decrease concentration |
|
|
Dosing: |
|
|
|
Start dose |
2mg |
2mg |
|
Titration increment |
2mg |
2mg |
|
Titration interval |
2 weeks |
1 week |
Hepatic disease
• Metabolism was different – up in some cases, down in others
• Overall steady state will probably take longer to achieve and so slower titration may be appropriate
Renal disease
• Renal impairment not expected to have a significant effect
• However not well studied in this population and should probably be avoided in severe renal impairment
Pregnancy
• Some effects in animals at high doses
• Australian Category B3
• Vazquez et al. Perampanel and pregnancy, Epilepsia 2021
o 48 pregnancies went to term – no abnormalities
o 28 did not reach term – reasons not all known - no evidence of higher than expected foetal malformation
Effectiveness
• 3 major phase III trials – all in patients with refractory focal epilepsy:
o 304
o 305
o 306
• All with slightly different doses and populations
Pooled phase III results
• Population
o Age 33-36yrs
o Caucasian 60-80%
o Epelepsy diagnosis ~20years
o Refractory seizures – average 9-14/28 days
o 70% secondary generalised
o >80% had failed 2 or more AEDs
o All were on at least on other AED, most were on 2 or 3
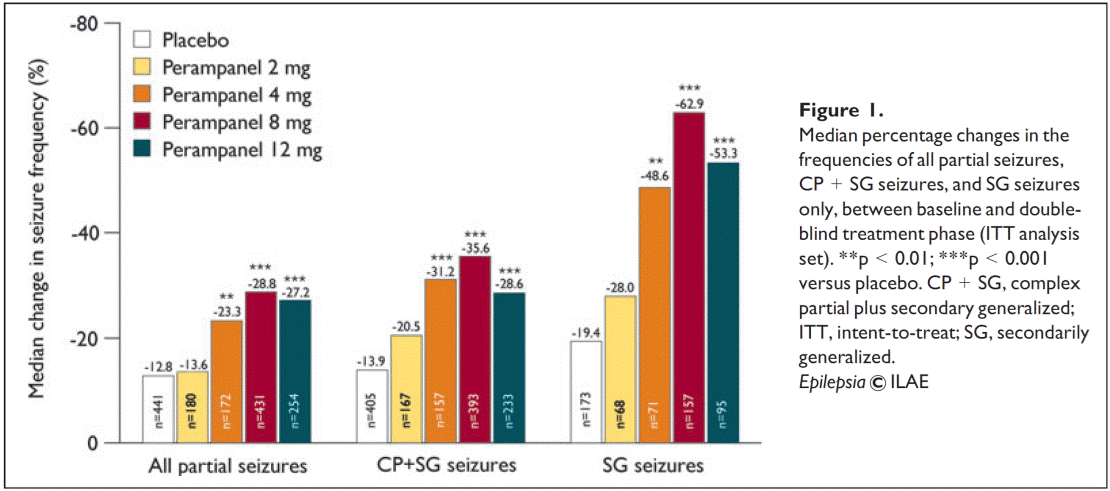

Treatment of Genetic Generalised Epilepsy
• French et al. Neurology 2015
• RCT 164 patients
• GGE with GTCS
• Titrated to 8mg daily
• Median % seizure change -38% vs -77%
• 50% responder rate 40% vs 64%
• Seizure freedom 12% vs 31%
• Similar adverse events to previous studies
• No exacerbation of other seizure types.
Side effects
Withdrawal due to adverse events:
|
Dose |
% Withdrawal |
|
Placebo |
4.8% |
|
2 |
6.7% |
|
4 |
2.9% |
|
8 |
7.7% |
|
12 |
19.2% |
|
Overall |
9.5% |
Adverse effects (pooled phase 3)
|
|
Placebo |
4mg |
8mg |
12mg |
|
|
Dizziness |
9 |
16 |
32 |
43 |
|
|
Headache |
11 |
11 |
11 |
13 |
NS |
|
Somnolence |
7 |
9 |
16 |
18 |
|
|
Fatigue |
5 |
8 |
8 |
12 |
|
|
Irritability |
3 |
4 |
7 |
12 |
|
|
Nausea |
5 |
3 |
6 |
8 |
|
|
Falls |
3 |
2 |
5 |
10 |
|
|
Ataxia |
0 |
0 |
5 |
3 |
|
|
Psychiatric: |
|
|
|
|
|
|
Aggression |
0.5 |
0.6 |
1.6 |
3.1 |
|
|
Anger |
0.2 |
0 |
1.2 |
2.7 |
|
|
Depression |
1.6 |
0.6 |
0.7 |
2.4 |
|
|
Anxiety |
1.1 |
1.7 |
3.0 |
3.5 |
|
• Psychiatric adverse events, in particular aggression, were higher in adolescent subgroup (small numbers).
Average weight gain of ~1.8 kg on perampanel - ~3-5% of patient report significant weight gain
Rufinamide
Dosing

Side Effects
Increased Liver Function Tests
Suggested criteria for stopping AED:
· ALT >10xULN on
one occasion
· ALT >3xULN
persistently (Over 3-6 months)
· Bilirubin >40
or >2xULN
·
Long term risks:
Cardiovascular risk with enzyme inducing drugs
• Large retrospective cohort study - (Josephson CB et al. Association of enzyme-inducing antiseizure drug use with long-term cardiovascular disease. JAMA Neurol 2021 Nov; 78:1367.)
o Hazzard ratio for cardiovascular disease in whole group 1.21. This drops to 1.06 if stricter definition of epilepsy is used.
o Difference became apparent after ~10 years
o Dose dependent