Stiff-Person
Syndrome and PERM
Anti-GABAB
Receptor Antibodies
Summary
|
|
|
|
|
|
|
|
|
NMDAR |
|
|
|
VGKC |
|
|
|
- LgI1 |
|
|
|
- CASPR2 |
|
|
|
GABAbR |
|
|
|
GABAaR |
|
|
|
GlyR |
|
|
|
mGluR5, mGluR1 |
|
|
|
IgLON5 |
|
|
|
Neurexin3a |
|
|
|
D2R |
|
|
|
DPPX |
|
|
|
GAD |
|
|
|
|
|
|
|
|
|
|
|
Hu, Ri |
|
|
|
Ma1/Ma2 |
|
|
|
PCA2 |
|
|
|
Yo |
|
|
|
CV2/CRMP5 |
|
|
|
Recoverin |
|
|
|
AGNA |
|
|
|
DNER |
|
|
|
ANNA 3 |
|
|
|
ZIC 4 |
|
|
|
Amphiphysin |
|
|
|
|
|
|
Anti-NMDAR encephalitis
• First recognised as a specific autoimmune condition in 2005
• ‘Four young women who developed acute psychiatric symptoms, seizures, memory deficits, decreased level of consciousness and central hypoventilation associated with ovarian teratoma and CSF inflammatory abnormalities’
Epidemiology
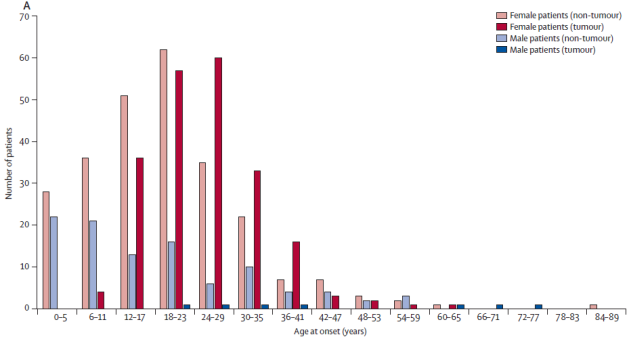
• Women ~80%
• Age
o Very rare after age 40
o Peak 18-30years
o Patients with tumour on average a bit older
Tumour association
• Tumours found in ~50% of patients – overall
• Adult women - 60%
• Children - 10%
• Men - 5% (testicular germ-cell, SCLC, lymphoma)
• Mostly ovarian teratomas (>90%)
• Express NMDAR (100% of 25 tumours studied)
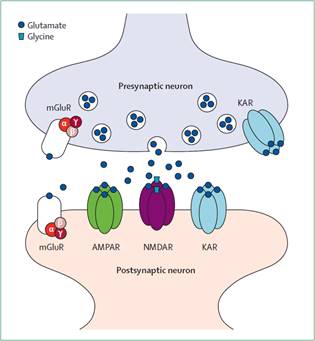
Pathogenesis
• Glutamate- principal excitatory neurotransmitter in CNS
• There are 3 ionotrophic glutamate receptors
o AMPA
o NMDA (N-methyl-D-aspartate)
o KA
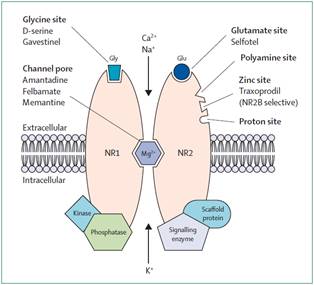
• NMDA receptor antagonists
o Ketamine and PCP - Hallucinations, paranoia, sedation, seizures
• Excess activation of neurons by excitatory neurotransmitters – particularly glutamate implicated in neuronal death – excitotoxicity
• Autoantibodies against NMDAr (subunit NR1)
• Internalisation of NMDA receptor and reduced synaptic density of NMDA receptor
Clinical Features
• Tend to have a usual course of onset and progression:
|
Prodrome (?70%) <2 weeks prior |
Headache, fever Nausea, vomiting, diarrhoea URTI |
|
Psychiatric |
Anxiety, fear, delusions Mania, Paranoia |
|
Cognitive |
Short-term memory loss Language impairment |
|
Seizures |
Complex partial |
|
Altered consciousness |
Catatonia |
|
Abnormal movements |
Oro-lingual-facial dyskinesia Dystonia |
|
Autonomic instability |
Hyperthermia, tachycardia, hypotension |
|
Hypoventilation |
Full or nocturnal ventilation |
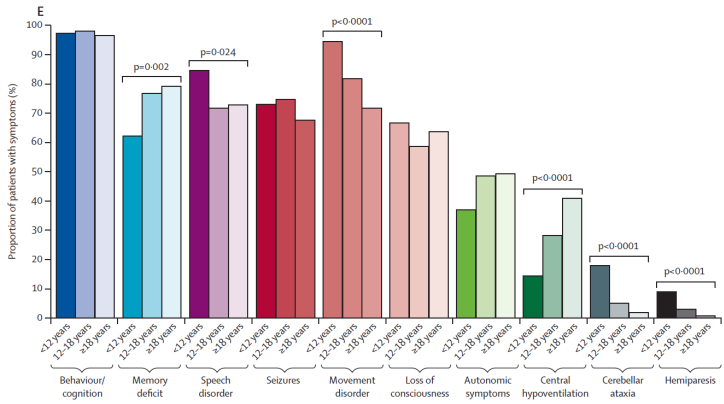
Symptoms at 1 month post onset (Lancet Neurol 2013)
Psychosis
• Psychosis (Zandi et al. J Neurol 2011)
o Prospective study of patients presenting with new-onset psychosis in the UK.
o 46 patients – 2 positive for NMDAR Abs, 1 VGKC Ab’s
o ~ 6.5% with potentially treatable cause
Epilepsy
• Arch Neurol 2009 – Dalmau, Vincent, Bien
• 847 women with epilepsy presenting to tertiary hospital in Germany.
• 19 had ‘unexplained’, new-onset epilepsy
• 5 had NMDAR antibodies
• 0.6% of all epilepsy in women
• 26% of ‘unexplained’, new-onset epilepsy in women
• Clinical Course
• Natural history highly variable
• 4 cases diagnosed prior to role for immunotherapy known
• Gradual recovery – mean 7months in hospital, 2 patients required >3 yrs to fully recover.
• Mortality 4% (mixed group with variable treatment)
•
Relapses
• 25% of untreated patients at 2 years
• 7-10% of treated patients at 2 years
• Up to 7yrs post initial episode
• Risk factors for relapse
o No immunotherapy at first episode
o No tumour
Investigations
Imaging
• MRI
o Abnormal in 35-50%
o A range of transient non-specific changes +/- subtle contrast enhancement
• PET/SPECT
o Variable, multi-focal cortical and subcortical changes
EEG
• Abnormal in most – but non-specific
• Generalised slowing ~80%
• Epileptiform discharges ~50%
• Seizures (occasional focal status)
• Extreme delta brush (possibly quite specific) – delta waves with brush like beta on the crests of the delta waves. Present in ~30% of adults with disease.
CSF analysis
• Initially abnormal in ~80%, becomes abnormal in most
• Mild/Moderate lymphocytic pleocytosis 70-90%
• Normal or mildly increased protein ~30%
• Oligoclonal bands ~60%
• NMDAr Abs 100%**
Brain biopsy
• not diagnostic
• Normal or non-specific inflammatory changes
NMDAr antibody testing
• Cell culture based assay:
o Perth, Pathwest, cost ~$21/test
o Can do CSF and serum
o Could do a semi-quantitative titre on special request
• Sensitivity - Good in acute disease
o ?delayed presentation or retrospective diagnosis
• Specificity - Probably excellent
o Some low level elevation in neurological control patients – particularly in serum
o It is probable that previous, asymptomatic, exposure to HSV may result in NMDA antibodies in serum
• 10% are found in CSF only
Differential diagnosis
“Encephalitis” - Confusion/amnesia/altered conscious state +/- seizures
Infectious (HSV, VZV, JE, TB…)
Seizures –e.g. temporal lobe epilepsy
Drugs/Toxins
Demyelinating - ADEM
Autoimmune
Traditional Paraneoplastic (‘limbic encephalitis’)
Hashimoto’s encephalopathy
‘New channel-opathies’ – NMDA, VGKC
NMDA’s cousins…
• AMPAR antibody encephalitis
• Presenting as limbic encephalitis
• Female patients with a variety of tumours
• Respond well to tumour treatment and immunotherapy
• NMDA’s cousins…
• Anti –VGKC antibody disease
• Seizures – faciobrachial
• Limbic encephalitis (Dense amnesia, confusion, hallucinations, depression)
• Hyponatraemia
• No major tumour association; respond to immunotherapy
• LGI1 and CASPR2 antigens
Management
Investigate for tumour
• Investigation for and removal of teratoma
• ? Presence of micro-tumour – prophylactic oophrectomy
• Tumour surveillance 1-2years
Immunomodulatory treatment
First Line:
• IVIG/plasma exchange + Methylprednisolone (5 doses of each)
• Evaluate after ~2 weeks – if response then consider repeating treatment after 1 month
If no response 2nd line therapies:
• Cyclophosphamide 750mg/m2 – monthly for 6 months
• and Rituximab 375mg/m2 weekly for 4 weeks
• Check for B-cell depletion at 2 weeks after Rituximab dose – if not sufficient give a further 2 doses
• If less than 16 years old start with rituximab only and see if cyclophosphamide can be avoided.
Long term immunosuppression
• ? Azathioprine, Mycophenolate
• No evidence
Psychiatric/behavioural
• Issues of competence and safety
• Will often need periods of chemical or physical restraint
• No evidence regarding specific anti-psychotic or sedative agents
• What is the role of ECT in patients who don’t rapidly recover with immune therapy? (case report of one patient with a good response)
Prognosis
Outcomes with treatment
~75% Good outcome
~21% Ongoing disability
4% mortality
Better prognosis if:
Paraneoplastic cause (and tumour removed)
Immunotherapy early (< 40days from onset)
Use of more combination immunotherapy (rather than steroids alone)
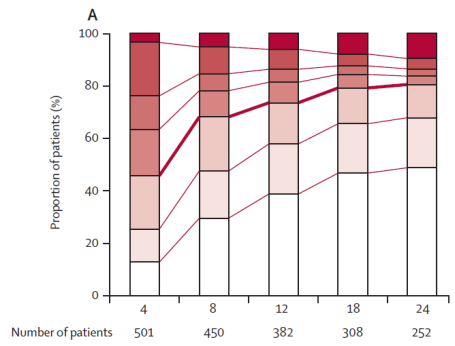
Prognosis (mRS scale) x-axis is months from diagnosis
References
Dalmau J et al, Clinical experience and laboratory investigations in patients with antiNMDAR encephalitis, Lancet Neurol 2011; 10: 63-74
Bataller L et al, Autoimmune limbic encephalitis in 39 patients: Immunophenotypes and outcomes, JNNP published online 15 Sep 2006 doi:10.1136/jnnp.2006.100644
Florance N et al, AntiNMDAR encephalitis in children and adolescents, Ann Neurol 2009; 66: 11-18
Pruss et al, Retrospective analysis of NMDA receptor antiboides in encephalitis of unknown origin, Neurology 2010; 75:1735-39
Dalmau et al, Anti-NMDA-receptor encephalitis: case series and analysis of the effects of antibodies, Lancet Neurol 2008; 7:1091-98
Vincent A and C Bien, Anti-NMDA-receptor encephalitis:a cause of psychiatric, seizure and movement disorders in young adults, Lancet Neurol 2008; 7:1074-5
Honnorat J, Autoimmune limbic encephalitis;An expanding concept, Lancet Neurol 2009; 9:25-25
Vedeler CA and A Storstein, Autoimmune limbic encephalitis, Acta Neurol Scand 2009; 120 (Suppl 189):63-67
Kalia LV, S Kalia and M Salter, NMDA receptors in clinical neurology:excitory times ahead, Lancet Neurol 2008; 7:742-55
Vincent A, S Irani and B Lang, The growing recognition of immunotherapy responsive seizure disorders with autoantibodies with specific neuronal proteins, Current opinion in Neurology 2010; 23:144-150
Gabilondo et al. Analysis of relapses in anti-NMDAR encephalitis. Neurology 2011; 77:996-999
Anti-VGKC Encephalitis
Diagnosis
· 85% of Anti-VGKC are ‘false positive’
· LGi1 and CASPER2 (subset of Anti-VGKC) are highly specific for disease
Imaging
50% of patients found to have T1 hyperintensity in the caudate head
Anti-GAD
Stiff-Person Syndrome and PERM
PERM
Antibodies:
DPPX (Dipeptidyl peptidase-like protein 6)
· Extracellular component of neuronal Kv4.2 potassium channels
· Neurology 2014 82(17) pg1521
Anti-GABAB Receptor Antibodies
• Only a few small case reports
• Lennon’s Mayo clinic group (Jerffery wt al. Neurology 2013) found 17 patients
o 0.2% of samples sent for autoimmune encephalopathy
o 10% of samples which had previously been classified as unknown autoimmune encephalopathy (based on hippocampal staining)
o 1.3% of samples which contained other SCLC antibodies
o The majority of patients had limbic encephalitis
o The majority had SCLC
• Dalmau’s group (Hoftberger et al., Neurology 2013) screened serum and CSF from ~9000 patients referred with possible autoimmune encephalitis or paraneoplastic syndromes.
o 20 patients were identified
o 10 had SCLC, 10 had no cancer
o The majority presented with a limbic encephalitis
o One patient had ataxia, one opsoclonus-myoclonus
o 15 of the patients had some response to immunotherapy – more so in the non-cancer group.
o One patient had NMDARA (and more psychiatric symptoms), one patient had anti-GAD (and had more seizures).
o The same paper suggested the anti-bodies were causing blockade of the receptor (rather than internalisation)
Hoftberger et al. 2013 Neurology Vol 81 pg1500
Anti-DPPX
• Stiff-Person syndrome and PERM
• Case reports of