Cannabis for epilepsy
•
•
Epilepsy trials
Tuberous Sclerosis GWEP1521 (2019) – unpublished
• CBD
25mg/kg vs 50mg/kg vs placebo
• ~75pts/group
• Median
sz reduction
o
27% vs 49% vs 48% (i.e.
~22% reduction relative to placebo)
• Adverse
effects (25mg/kg group)
o
Leading to withdrawal ~8% (relative to placebo)
o
Diarrhoea 6%
o
Somnolence 4%
o
Increased liver enzymes ~11%
o
Vomiting 7%
o All higher in the 50mg/kg group
RCTs as per 2020:
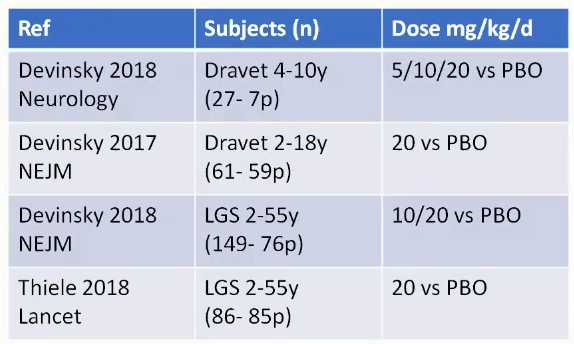
• Pooled median seizure reduction ~19%
• 50% reduction – RR 1.8
• No dosing efficacy difference
• GI Side effects – OR 1.5
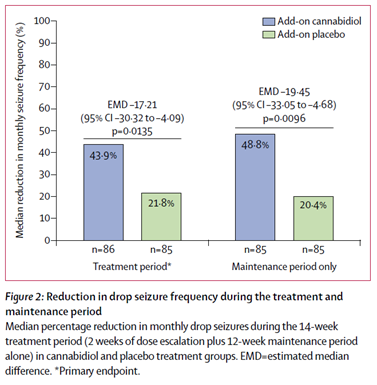
Thiele 2018 – GWPCARE4
• 2-55yrs old
o ~1/3 adults
• LGS
• 85 placebo vs 86 cannabidiol
• 2.5mg/kg increasing every 2 weeks up to 20mg/kg
• Mean seizure reduction - 22%
• Seizure free 3 (3.5%) vs 0
BELIEVE 1 (2019)
·
Zygel in developmental
and epileptic encephalopathies
·
Phase 2 trial
·
44% reduction in seizures ?but
open label
Negative trials
CBD-V GW 2017-2019 – unpublished
• No difference between placebo and Rx
ZYNERBA – STAR trial - unpublished
• Australian trial 2018
• Negative result