Dementia
and cognitive impairment
Memory
Short term memory
• “Working memory”
• Normal person can hold ~7 bits of information
• Dependent on attention
Long term memory
• Declarative
o Semantic
- Knowledge about the world, generic (vocabulary is large part)
- Cortical association areas
o Episodic
- recollection of unique personal experiences (e.g. remembering a holiday)
- Medial temporal lobe and neocortex
• Nondeclarative
o Procedural
- E.g. how to swing a golf club
- Striatum
o Conditioning
o Nonassociative learning
Definitions
Subjective Cognitive Impairment (SCI)
• Subjective concern about decline in cognitive abilities with out objective evidence
Mild Cognitive Impairment (MCI)
• Objective decline greater than expected for age
• Does not interfere with activities of daily life
Functional Cognitive Impairment (FCD)
• Subjective symptoms of cognitive impairment
• Evidence of internal inconsistency
• Objective testing may or be positive or negative.
Dementia
• Objective decline greater than expected for age
• Interferes with activities of daily life
Dementia
overview
DDX
Also see: Dementia - Rapidly Progressive
Most Common Causes of Dementia
Alzheimer's
disease
Vascular dementia
Multi-infarct
Diffuse white matter disease
(Binswanger's)
Alcoholisma
Parkinson's
disease
Drug/medication intoxicationa
Less Common Causes of Dementia
Vitamin
deficiencies
Thiamine (B1):
Wernicke's encephalopathya
B12 (pernicious anemia)a
Nicotinic acid (pellagra)a
Endocrine and
other organ failure
Hypothyroidisma
Adrenal insufficiency and
Cushing's syndromea
Hypo- and hyperparathyroidisma
Renal failurea
Liver failurea
Pulmonary failurea
Chronic infections
HIV
Neurosyphilisa
Papovavirus
(progressive multifocal leukoencephalopathy)
Prion (Creutzfeldt-Jakob and
Gerstmann-Sträussler-Scheinker
diseases)
Tuberculosis, fungal, and protozoala
Whipple's diseasea
Head trauma and
diffuse brain damage
Dementia pugilistica
Chronic subdural hematomaa
Postanoxia
Postencephalitis
Normal-pressure hydrocephalusa
Neoplastic
Primary brain tumora
Metastatic brain tumora
Paraneoplastic limbic
encephalitis
Toxic disorders
Drug, medication, and narcotic poisoninga
Heavy metal intoxicationa
Dialysis dementia (aluminum)
Organic toxins
Psychiatric
Depression (pseudodementia)a
Schizophreniaa
Conversion reactiona
Degenerative
disorders
Huntington's disease
Pick's disease
Dementia with Lewy bodies
Progressive supranuclear palsy
(Steel-Richardson syndrome)
Multisystem degeneration (Shy-Drager syndrome)
Hereditary ataxias
(some forms)
Motor neuron
disease [amyotrophic lateral sclerosis (ALS); some forms]
Frontotemporal dementia
Cortical basal degeneration
Multiple sclerosis
Adult Down's syndrome with
Alzheimer's
ALS–Parkinson's–Dementia
complex of
Miscellaneous
Sarcoidosisa
Vasculitisa
CADASIL etc
Acute intermittent porphyriaa
Recurrent nonconvulsive seizuresa
Additional
conditions in children or adolescents
Hallervorden-Spatz
disease
Subacute sclerosing
panencephalitis
Metabolic
disorders (e.g., Wilson's and Leigh's diseases, leukodystrophies, lipid storage
diseases, mitochondrial mutations)
Comparison of Dementia
Types
|
Disease |
First
Symptom |
Mental
Status |
Neuropsychiatry
|
Neurology
|
Imaging |
|
AD |
Memory loss |
Episodic memory loss |
Initially normal |
Initially normal |
Entorhinal cortex and hippocampal atrophy |
|
FTD |
Apathy; poor judgment/insight,
speech/language; hyperorality |
Frontal/executive, language; spares drawing
|
Apathy, disinhibition, hyperorality,
euphoria, depression |
Due to PSP/CBD overlap; vertical gaze
palsy, axial rigidity, dystonia, alien hand |
Frontal and/or temporal atrophy; spares
posterior parietal lobe |
|
DLB |
Visual hallucinations, REM sleep disorder,
delirium, Capgras' syndrome, parkinsonism |
Drawing and frontal/executive; spares
memory; delirium prone |
Visual hallucinations, depression, sleep
disorder, delusions |
Parkinsonism |
Posterior parietal atrophy; hippocampi
larger than in AD |
|
CJD |
Dementia, mood, anxiety, movement disorders |
Variable, frontal/executive, focal
cortical, memory |
Depression, anxiety |
Myoclonus, rigidity, parkinsonism |
Cortical ribboning and basal ganglia or
thalamus hyperintensity on diffusion/flare MRI |
|
Vascular |
Often but not always sudden; variable;
apathy, falls, focal weakness |
Frontal/executive, cognitive slowing; can
spare memory |
Apathy, delusions, anxiety |
Usually motor slowing, spasticity; can be
normal |
Cortical and/or subcortical infarctions,
confluent white matter disease |
Prevalence
• Alzheimers – 65%
• Vascular – 5%
• Vascular + AD – 10%
• Lewy bodies – 7%
• AD + DLB – 5%
• Other – 8%
o Fronto-temporal
o CJD
Diagnosis
Criteria
1.
Development of multiple cognitive impairments
including memory impairment and at least one:
a.
Aphasia, apraxia, agnosia
b.
Disturbance of executive functioning
2.
Sufficiently severe to cause impairment in
occupational and social functioning and represent a decline
3.
Chronic (progressive or static)
4.
Diagnosis should not be made during delerium
Investigations
• Routine
o FBC/U+E
o LFT
o TFTs
o B12
o RPR
o CT
• Optional
o Ammonia
o Heavy metals
o HIV
o LP (RPR, CJD)
o EEG (usually only useful for CJD)
o Cerebral angiography (vasculitis)
o Brain biopsy
• Consider exacerbating factors
o Depression
o Carer stress
o Drugs
o Seizures
o Insomnia
o Agitation
Mild Cognitive impairment
• 50% progress to dementia in 5 years – 10%/year
Treatment
• No proven intervention to prevent progression.
• Vitamin E – no effect
• Donepezil
o Small benefit at one year but none by 3 years in one trial
o Other trials negative
• Galantamine
o No effect and potential small increase in mortality
• HTN
o Treatment of HTN in general is associated with lower risk of dementia however no trials done specifically in MCI
• Physical activity
Functional Cognitive Disorder
Diagnostic criteria
1. One or more symptoms of impaired cognitive function
2. Clinical evidence of internal inconsistency
3. Not better explained by another medical or psychiatric disorder
4. Cause clinically significant distress or impairment in one or more functional domains or warrants medical evaluation.
Internal inconsistency
• The ability to perform a task well at certain times, but with significantly impaired ability at other times, particularly when the task is the focus of attention.
• Individual components required to execute a task are intact, however there is difficulty engaging them at the appropriate intensity or duration on demand.
• Positive evidence can be demonstrated by any of the following:
o Where subjectively-reported cognitive difficulties, and/or low standardized cognitive test scores, directly contrast with:
- (a) Conversational abilities observed during interview
- (b) Reported activities, such as being involved in a cognitively demanding occupation; or difficulties only occurring in particular situations.
- (c) Collateral history suggesting concern is significantly higher in the individual than their supporter (including the ‘attended alone’ sign)
o (ii) Specific patterns within neuropsychological testing that indicate cognitive processes performing better when accessed less explicitly, e.g. greater ability in delayed recall than initial registration of information.
• A patient tendency to give approximate answers could also be used as an example of internal inconsistency
• (Ganser's syndrome is a rare and controversial condition, whose main and most striking feature is the production of approximate answers (or near misses) to very simple questions.)
Reference: Ball HA, McWhirter L, Ballard C, et al. Functional
cognitive disorder: dementia's blind spot. Brain. 2020;143(10):2895-2903. doi:10.1093/brain/awaa224
(https://pubmed.ncbi.nlm.nih.gov/32791521/
)
Alzheimer’s
Disease
Epidemiology
• Most
common form of dementia (50-60%)
• F>M (small difference)
• Prevalence :
o <60yrs - <1%
o >85yrs ~25%
Risk factors
• Age (strongest risk factor – risk doubles every 5 years after 65years)
• Family history (3-5x risk in first degree relatives)
• Down’s syndrome (APP gene on chromosome 21)
• Mild cognitive impairment
• Decreased ‘reserve capacity’ of the brain
• Low mental ability in early life
• Reduced mental and physical activity during later life
• Head injury could also be a RF.
• Cardiovascular risk factors
o HTN, DM, Obesity, Homocysteine
Protective factors
• Education
• Moderate alcohol consumption
• ?HRT
• ?NSAIDS
Genetics
• Some small genetic familial groups (<0.1%)
o Mutations in APP and Presenillin 1 and 2.
o Presenillin-1 and -2
- Presenilin 1 results in early onset disease (age ~45) and more rapidly progressive, as does Presenillin-2 (age ~53)
- Account for ~?10-50% of early onset AD
- Autosomal dominant
- Multiple different mutations
o APP mutations – accounts for only a small proportion of early-onset disease. Multiple different mutations.
• Genetic component of sporadic disease thought to be related to APOe4 allele
o Heterozygotes 3x risk
o Homozygotes 15x risk
o Main effect is lowering the age of disease onset (one decade per allele)
o Frequency in Australia ~14% (26% heterozygotes, 1-2% homozygote)
Pathology
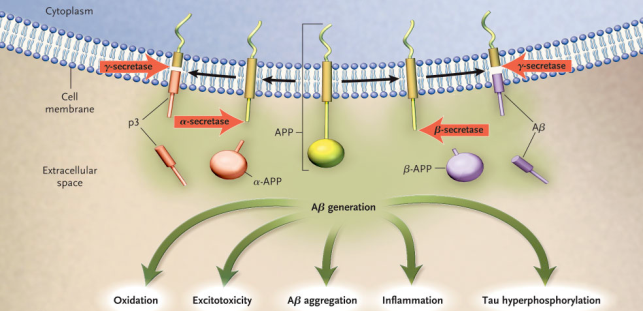
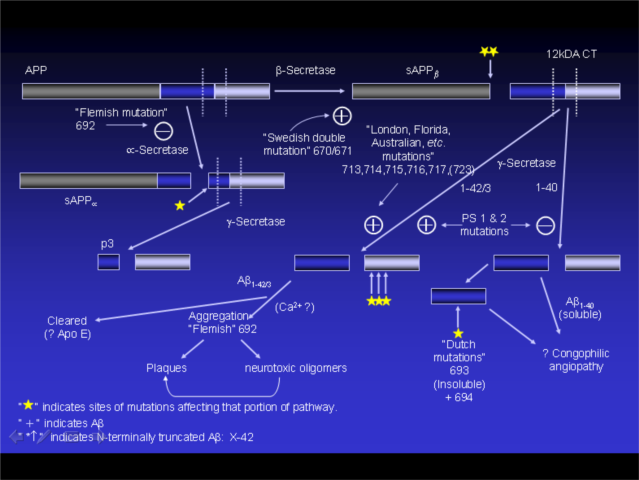
• Neuritic plaques (extracellular)
o Amyloid beta is major plaque component
o Produced from APP (amyloid precursor protein) cleaved via a number of enzymes in which Presenillin is involved
o ABeta oligomers are toxic, the actual plaque is not
o Imbalance between production and clearance thought to result in disease
o APP is found on chromosome 21 – people with down syndrome are prone to early onset of AD type illness probably as a result of extra APP
o APP is cleaved in two steps – alpha or beta secretase followed by gamma secretase
o If alpha cleaves then non-toxic metabolities
o If beta secretase cleaves first then gamma sectretase produces either AB (1-40) non-toxic or the toxic AB (1-42)
• Neurofibrillary tangles (intracellular)
o Hyperphosphorylated tau protein
o Tau normally binds to microtubules and promotes there assembly and stability
o Phosphorylation results in disassembly of the tubules and impaired transport.
o Probably not ‘pathogenic’ but better marker of disease severity than plaques.
Genetic Forms
• Early onset
o Mutations in genes involved in AB1-42 production
o APP mutations predispose to beta-secretase cleavage
o Presenillin 1 and 2 promote gamma-secreatase to produce AB(1-42)
• Late onset genetic
o Unknown proportion of AD ?10%
o ApoE e4 allele (see above)
o ApoJ – is newly identified potential RF
o Many other genes have some association, combined effect significant.
Clinical features
• Slowly
progressive
• Progressive memory impairment
o episodic memory
o Majority present with this however 20% present with other complaint
• Instrumental signs
o Aphasia
o Apraxia
o Agnosia
• General cognitive symptoms
o Impaired judgement
o Decision-making
o Orientation
Approximately 10% of AD patients develop Capgras' syndrome,
believing that a caregiver has been replaced by an impostor.
3 phases:
• Pre-clinical neurodegeneration (~20yrs)
• Mild cognitive impairment (~5 yrs)
o >1.5 SD below normal on memory testing.
o Other cognitive domains unaffected
o Function unaffected
o ~50% of patients with MCI progress to AD in 5years
o Some patients remain stable in long term with minimal or no progression
• Alzheimers disease
Progression
• Typical progression of cognitive impairment:
o Episodic
o Semantic
o Executive
• Matches pathological progression (mesial temporal – temporal neocortex – other cortex)
Diagnosis
• Memory
testing
• Exclusion of other forms of dementia
• NINCDS criteria – sensitive but not very specific, used more for research
• DSM-IV – more specific, less sensitive
• Diagnosis usually made by clinical judgement
McKhann criteria
• Definite
o Pathologically confirmed (Plaques and tangles)
• Probable
o Dementia
o Memory impairment plus at least one other cognitive domain
o Other causes excluded
• Possible
o Atypical course
Revised (McKann) 2007 criteria
• Core features
o Gradual and progressive memory progression over >6 months
o Objective evidence of impaired episodic memory with impaired encoding (not just retrieval) – (i.e. cannot get recall items even with prompting)
• Supportive features
o One or more of the following biomarkers:
- Medial temporal lobe atrophy on MRI
- Abnormal CSF tau: AB ratio
- Specific pattern on PET
- Proven causative AD mutation in family
Experimental investigations
• MRI – hippocampal atrophy more indicative of alzheimer’s
o cingulate atrophy
• PET – various modalities show promise, decreased metabolism in certain regions
o FDG PET – shows bilateral tempoparietal hypoperfusion/hypometabolism
o PIB PET – images amyloid
• LP biomarkers
o Dec CSF AB/tau ratio
Treatment
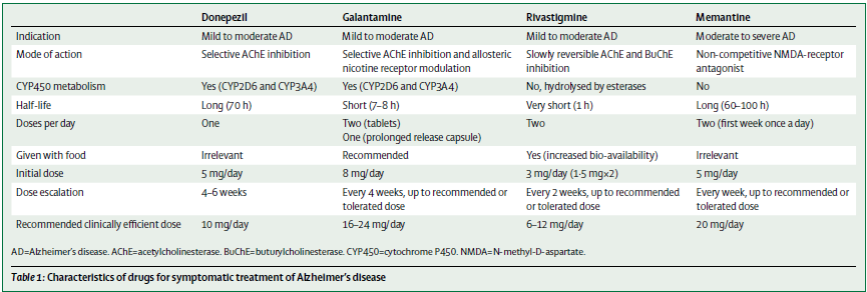
Acetylcholinesterase inhibitors
• Drugs
o Donepezil (Aricept)
o Galantamine (Reminyl)
- May have more GI effects
o Rivastigmine (Exelon)
- Available in a patch
• Centrally acting AChE-inhibitors with variable amounts of peripheral effect.
• Work on theory that loss of cholinergic neurones in basal forebrain nuclei causes decreased Ach mediated stimulation of hippocampus and neocortex.
• Severity of dementia
o Benefit in mild to moderate disease
o ? some benefit in severe
o No benefit in mild cognitive impairment
• PBS
o Mild-mod severe AD (MMSE >10 or equivalent)
o Need to show 2 point improvement in MMSE
• Overall mean improvement in MMSE varies significantly depending on study (0.8 and 1.4 quoted for MMSE and ADAS-Cog 2.7)
• At least may help keep MMSE stable for ~1year (as apposed to 2-3 point drop in controls)
• Some evidence of benefit out to 2 and perhaps 5 years
• Benefits in terms of ADL’s, and time to institutionalisation marginal/non-significant.
• SE: Cholinergic - Mainly GI symptoms
o Diarrhoea, nausea
o Increased urinary frequency
o Weight loss
o Insomnia (vivid dreams)
Memantine
• Noncompetitive NMDA antagonist
o excessive NMDA stimulation can result in excitotoxicity
o Interaction with glutamate producing neurones
• Study in NEJM 2003 in mod to severe dementia (MMSE 3-14) showed stabilisation on some functional scales
• Minimal side effects
• Combination with AChE-Inhibitors (vs ACHE-I alone) has been trialled with success
• PBS
o Need MMSE 10-14
• SE: can lower seizure threshold, non-specific – drowsiness, dizziness etc.
• Medications with some evidence for prevention:
o NSAIDS
o Statins
o Oestrogen (in HRT)
• Other disease prevention
o Prevention of HTN has some benefit
o Vitamin E
- Mixed trial results, some mildly positive for delaying clinical progression
- Concern about cardiac toxicity at high doses
o Selegiline (MAO inhibitor) some mixed evidence of effect.
Vascular
Dementia
• Risk Factors
o Hx of CVA increase risk x9
• Types:
o Multi-infarct dementia
o Diffuse white matter disease (Biswanger’s disease)
o Cerebral amyloid angiopathy
o Rare:
- CADASIL (Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy) (see CVA topic)
- Metachromatic leukodystrophy
- PML
- Mitochondrial disorders (MELAS, MERRF)
Clinical
• May be a step-wise decline
• May be focal neurological signs (to help differentiate from AD)
• Behavioural change
• Executive function changes
• Memory impairment
o Compared with AD - can lay down memory but not recall (prompting may help recall)
• Early urinary incontinence may be a sign
• Can co-exist with AD
Treatment
• Worse prognosis than AD
• Treat HTN may help prevent cognitive decline
• Aspirin
o No evidence of significant effect
• AChE-Inhibitors
o Donepezil and Galantamine have been studied and small benefits shown (not in functional assessments however)
Frontotemporal
Dementia
• Onset
35-75years, avg 60’s
• Male 2x more likely
• 20% familial
• 5% of all dementias, 40% of dementia under 60.
Types:
• Complex
and controversial area
• Clinically three main categories:
o Behavioural change predominant
o Semantic dementia
o Progressive non-fluent aphasia
• Pick’s disease is a subtype in which the classic pathological changes of Pick bodies are seen
• Overlap syndromes with ALS, PSP, CBD can also be defined
Pathology
• Majority
have tau inclusion bodies
• If these stain with silver stain they are referred to as Pick bodies
• Ubiquitin inclusion bodies may also occur
•
Serotonergic deficiency has been observed
Clinical
• Behavioural
variant
o (sometimes called frontal variant or classic FTD/Pick’s disease)
o Personality change
o Poor insight into condition
o Loss of social awareness
o Stereotyped or ritual behaviours
o Mental rigidity
o Executive function changes
o Memory impairment rare (<10%), visual and spatial skills relatively preserved
o Compulsive behaviour
o Hyper-orality
• Progressive Non-fluent aphasia
o (Previously primary progressive aphasia)
o Word-finding difficulty with preserved understanding of words
o Relatively preserved insight
o Anomia
• Semantic dementia
o (Sometimes temporal variant of FTD)
o Problems naming and understanding words, relative preservation of episodic memory
o Relative normal fluency – use of semantic paraphasias
o Repetition usually normal
o Poor performance on semantic fluency (e.g. list of animals), reasonable performance on letter fluency (e.g. list of words beginning with F), compared to other two variants which have problems with both.
• Motor deficits – overlap syndromes with ALS, PSP, CBD
Imaging
MRI
• Symmetrical or asymmetrical atrophy of the frontal lobes and anterior temporal lobes
• Asymmetrical left frontal lobe loss (Brocca’s area)
o Associated with non-fluent aphasia phenotype
• Asymmetrical left anterior temporal lobe loss (more Wernike’s like)
o Associated with semantic dementia type
• Asymmetrical right sided atrophy
o Associated with behavioural alterations.
PET
• Hypometabolism on FDG PET can confirm the patterns noted on MRI
Treatment
• No specific treatment
• SSRIs sometimes used
• AChE-inhibitors
o Some evidence of improved behaviour aspects but overall no consistent benefit demonstrated.
Dementia
with Lewy Bodies
Epidemiology
• Increases
with age
• Generally from 60years, median 75years
• Accounts for 10-20% of dementia
• 0.7% of population over 65years
• Majority sporadic
• Family history a risk factor generally
• Some genetic families
o Triplication of alpha synuclein gene (SNCA)
DDX
• Parkinsons
disease with dementia
o Arbitrary classification - if PD symptoms have been present for 1year (?2year) prior to onset of dementia.
• Parkinson’s disease forming in someone with dementia
o occurs not infrequently due to prevalence of both conditions.
o Should be suspected if PD features occur late in disease when severe dementia is already evident.
• PSP
• CBD
• MSA
Pathology
• Presence
of Lewy bodies
• Dense lesion with surrounding halos
• Found in Substantia nigra and throughout the brain
• Composed largely of:
o Alpha-synuclein
- protein probably involved in vesicle transport
- reason for accumulation not well understood
o Ubiquitin
• Presence of neurofibriliary tangles and amyloid deposits can also occur similar to AD
• Density of Lewy bodies related to disease severity
Imaging
• MRI
o Relative preservation of medial temporal lobe/hippocampus
o More pronounced cortical atrophy
o MRI findings however not specific enough to be used to routinely differentiate the conditions.
• And rather randomly…..Low uptake on MIBG myocardial study
o Similar finding in PD but not Parkinson plus syndromes
• May be occipital hypoperfusion on SPECT and PET
Clinical
Cardinal features:
• Gradually progressive dementia (100%)
• Fluctuations in cognitive function (60-80%)
• Persistent, well-formed visual hallucinations (50-75%)
• Spontaneous motor features of parkinsonism (70-90%)
Other features:
• Repeated falls
• Syncope
• Autonomic dysfunction
• Sensitivity to neuroleptic medications delusions
• hallucinations in non visual modes
• REM sleep behaviour disorders
• Depression
• Attention and visuospatial impairment
Treatment
- Cholinesterase
Inhibitors
- The Ach
deficit is thought to be bigger in DLB than in AD
- These drugs
have been proven effective (although less studies than AD)
- Reduction in
anxiety, delusions and hallucinations.
·
Memantine
o
Small benefit in one study in some aspects however
worsening of symptoms in some patients.
- Antipsychotics
- Atypical
agents only should be used as the anti-dopamine effects of others may
worsen disease
- Caution
should be used
- Parkinson’s
symptoms
- Treatment
should be the same as for PD