MS Treatment – preventaive immunomodulatory treatments
Interferon
beta (Rebif, Plegridy, Avonex)
Teriflunomide (Aubagio, Teriflagio)
Daclizumab
(Zinbryta) - DISCONTINUED
Interferon beta
(Rebif, Plegridy, Avonex)
Efficacy
• 18-34% reduction in annualised relapse rate
•
Betaferon - Initial placebo controlled trial - Neurology. 1993;43(4):655
• INCOMIN trial – Avonex vs betaferon – favoured latter. Another Danish study found similar efficacy.
• PRISIMS trial (Rebif vs placebo)
• EVIDENCE trial (Neurology. 2002;59(10):1496) Rebif vs AVONEX – favoured Rebif
• In general terms the evidence for Avonex suggests it may be less effective on some parameters of treatment.
Adverse effects
Contraindications:
• Hypersensitivity to interferon
Adverse effects:
• Injection site reaction – 50% (reduce slightly over time)
• Influenza like symptoms – 40% (reduce to 25% over time)
• Myalgia/arthralgia
• The risk of depression has been raised – however appears similar to placebo group
Complications:
• Increased LFTs – occurs in up to 37%, severe in 1-2% - usually transient
• Decreased white cell count in ~7%
• Severe or symptomatic decreases in white cell count or platelet count have occurred very rarely
• Thrombotic microangiopathy has been noted rarely in case reports
• Seizures have noted in rare case reports
Pregnancy and breastfeeding
• No increase in major congenital malformations
• Safe with breastfeeding
Dosing
• Interferon beta – 1b (Betaferon) 0.25mg SC second daily
• Interferon beta – 1a
o Avonex - 30mcg IM weekly
o Plegridy (Pegelated) – SC every 2 weeks
- Titration – 1st dose 63mcg, 2nd dose 94mcg, ongoing dose 125mcg
• Interferon beta – 1a (Rebif) 22 or 44mcg SC three times/wk
Monitoring
• FBC/LFT ~ 6 weeks after starting and 6 monthly thereafter
Glatiramer (Copaxone)
• Mixture of random polymers from 4 amino acids
• Antigenically similar to myelin basic protein, distracts immune cells from destruction of myelin sheath
Efficacy
• Lowers relapse rate ~25% (1.19 vs 1.68), reduction in T2 lesions
• No evidence of preventing progression of disability
• Similar efficacy to IFN, similar tolerability, different SE profile (see BEYOND trial below)
Adverse effects
• Injection site reactions (22%)
o Redness, pain, pruritus, mass
• Lipoatrophy
• Immediate post injection reaction (2%)
o Transient chest pain, flushing, dyspnoea, palpitations/anxiety, constriction of the throat, urticaria.
o Can occur with any injection, random
• Lipoatrophy (0.5%)
• Skin necrosis (case reports only)
Pregnancy and breastfeeding
• No increase in major congenital malformations
• Safe in breastfeeding
Dosing
• 40mg SC 3x/week
Monitoring
• FBC/LFT 6 weeks after starting and yearly thereafter
Fingolimod (Gilenya)
Mechanism
• Analogue of Sphingosine 1-phosphate (S1P)
• Acts as a functional antagonist at S1P receptors
• S1P1 receptors are involved in trafficking of lymphocytes through lymph nodes – fingolimod prevents lymphocyte egress from lymph nodes.
• Fingolimod is non-specific and also bind to S1P 3, 4 and 5 receptors.
Efficacy - Trials
TRANSFORMS – NEJM 2010 362:402
• Fingolimod 1.25mg vs 0.5mg vs Interferon beta 1a (avonex) 30mcg
• Primary end point – Annual relapse rate
o 0.2 vs 0.16 vs 0.33
• Progression of sustained (>3months) disability
o Hardly any patients had progression of disability – no difference between any groups
FREEDOMS trial – NEJM 2010 362:387
• Fingolimod 1.25mg vs 0.5mg vs placebo
• Annualised relapse rate - 0.18 vs 0.18 vs 0.4
• Absence of disability progression at 2 years – 83.4 vs 82.3 vs 75.9 (significant)
Adverse effects and Contraindications
Contraindications
• Bradycardia or conduction abnormalities
• Other unstable heart disease
• Active or latent infections (including HBV, HCV, HIV)
• Severe liver dysfunction
• Lymphopenia
• Macula oedema
o ?previous uveitis
Adverse effects
• Muscle/back ache -
rare
• GIT – diarrhoea,
nausea – very rare
Complications
• Bradycardia
o
Only with first dose (or if after prolonged break
from medication)
o
Contraindicated in patients with heart block
o
Caution if on negatively chronotropic drugs
• Increased liver
function tests
o
Increased ALT ~7%
- Cease
treatment if >5x ULN, most LFTs return to normal despite continuing
treatment.
• Lymphopaenia
o Average lymphocyte count falls 75% (from 1.8 to ~?0.5)
o No increased risk of infection has been demonstrated with this fall – lymphocytes are still present, just not in circulation.
o Concern that very low count (<0.2) could result in increased infection risk
• Infections
o
Herpes virus infection
- Increased risk of
VZV infection and HSV
- Rare cases of
disseminated VZV and fulminant HSV encephalitis
o
Cryptococcal meningitis and other fungal
infections.
• PRES – rare cases reported ?association.
• Hypertension –
increase of 3mmHg over placebo
• Macular oedema
o
1/300 risk
o
Majority of cases within first 6 months
o
Majority of cases reversible
o
Biggest risk factor is previous uveitis. Uncontrolled DM also a risk factor.
• Excess skin cancer
risk
• PML
o Update report of Novartis database 2018 (Neurology 2018;90, pg 920)
o Risk of PML in patients treated with Fingolimod 0.069/1000pts
o Incidence 3.12/100,000 patient-years
o Not associated with lymphopaenia
o 14/15 pts had been treated for >2 years
Pregnancy and breastfeeding
• Some cases of congenital malformation - ?unclear if above background risk
o Advise patients to avoid in pregnancy
o Consider avoid pregnancy for 2 months post treatment cessation
• Breast feeding – transferred in breast milk – breast feeding not advised.
Pharmacology and Interactions
• Half life 6-9 days – steady state after ~1 months
• Complete elimination from the body ~2 months
• Caution with any negatively chronotrophic agents that may exacerbate bradycardia
• Ketaconazole can increase fingolimod levels
• Carbamazepine and other strong CYP450 inducers may result in reduced fingolimod levels
Dosing
• 0.5mg BD for all
• No need to dose adjust for renal or hepatic impairment
Pre-treatment screening
|
Test |
Reason |
|
BHCG
or urine pregnancy test |
Increased
risk of foetal malformation |
|
FBC |
Lymphopenia often occurs |
|
LFT |
Raised
LFTs can occur |
|
Hepatitis B and C |
Risk unknown, excluded from trials. |
|
VZV
IgG |
Disseminated
zoster has occurred. Vaccinate if
negative. |
|
HIV |
Risk unknown, excluded from trials. |
|
Quantiferon gold |
No
known increased risk. |
|
CXR (optional) |
Any evidence of TB or other infectious disease. Baseline. |
|
ECG |
Bradycardia
or heart block – worsened by fingolimod. |
|
Skin Check |
Possible increased risk of skin cancer. |
|
Ophthalmology
review or macula OCT |
Baseline
for macula oedema. |
|
Respiratory function tests (optional) |
Optional, there may be slight decrease in lung function. |
|
First
Dose cardiac monitoring |
All
patients, 6 hours (see below) |
•
Frist dose monitoring
o
Baseline ECG
o
Hourly Pulse and BP measurements
o
Perform ECG at 6 hours
o
If pulse <45bpm or lowest post dose value –
prolong monitoring until resolved
o
If QTc >470 (female) or >450 (male – observe
overnight and monitor for second dose
o
First dose monitoring should be repeated if
treatment discontinued:
- >/= 1 day in
first 2 weeks
- >7 days in week
3-4
- >2 weeks after
first month
• Vaccinations
should be completed 6 weeks prior to treatment.
Influenza vaccine can be given while patient is on medication, however may not be as effective.
Advice to patients
• Drug must be taken continuously – breaks in treatment may lead to bradycardia on restarting
• Watch for:
o Rash (particularly shingles)
o Jaundice
o Visual distortion (consider screening with Amsler grid)
• Vaccinations:
o Try and get vaccinations done prior to treatment
o Annual influenza vaccine encouraged
o Avoid live attenuated vaccines while on treatment
• Malignancy – ensure standard malignancy screening, including yearly skin check
• Women – avoid pregnancy, ensure adequate contraception, notify treating team immediately if pregnant
Siponimod (Mayzent)
Mechanism
• Analogue of Sphingosine 1-phosphate (S1P)
• Acts as a functional antagonist at S1P receptors
o Higher affinity at S1P-1 than fingolimod
o More specific than fingolimod (also bind to S1P 5 receptors)
• S1P1 receptors are involved in trafficking of lymphocytes through lymph nodes – siponimod prevents lymphocyte egress from lymph nodes.
Dosing
• 0.25mg titration over 5 days
• Ongoing dose:
o 2mg for standard metabolisers
o 1mg for poor metabolises
• PBS criteria:
o Previous diagnosis of RRMS
o Ambulatory
o Mild disability in at least 3 functional systems OR Moderate disability in at least 1 FS.
- (Functional systems: visual, brain stem, pyramidal, cerebellar, sensory, bowel/bladder and cerebral/cognitive systems)
o For continuing use: Must not show continuing progression of disability while on treatment
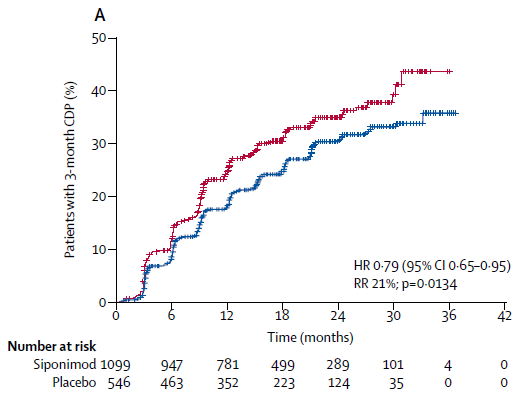
Efficacy – Trials
• EXPAND Trial
o Lancet 2018
o 18-60yrs old, diagnosis of SPMS, 1600pts
o Moderate to advanced disability (EDSS 3.0-6.5)
o EDSS progression in previous 2 years
o No evidence of relapse for 3 months (~22% had relapse in prior year and 35% in prior 2 years)
o Exclusion: Significant cardiac/pulmonary disease, uncontrolled DM, CYP2C9*3/*3 genotype
o 2mg vs placebo (titrated from 0.25mg to 2mg over 6 days)
o 21% had GAD enhancing lesions on T1 weighted imaging!
o Primary endpoint – confirmed disability progression at 3 months
- Sp 26% vs Placebo 32% (ARR 6%)
o Secondary endpoints
- 25ft walk test – no difference
- Annualised relapse rate – 0.07 vs 0.16 (HR 0.54)
- Patients with new T2 lesions – Sp 43% vs Placebo 63%
- Patients with new T1 lesion – 11% vs 33%
- Brain volumes better on treatment at 12 months, but no difference by 24 months
o The primary endpoint was non-significant in group without relapse in previous 2 years.
Adverse effects and contraindications
Contraindications:
• Cardiac/vascular disease
o Severe cardiac arrhythmia (particularly requiring medication)
o History of heart block (other than first degree)
o QTc >500ms
o History of symptomatic bradycardia or recurrent syncope
o History Bradycardia or conduction abnormalities
o In last 6 months - Ischaemic heart disease, stroke, heart failure
• Other cardiac disease – may consider treatment, however suggest cardiology review and first dose monitoring
• Severe untreated OSA
• Active or latent infections (including HBV, HCV, HIV)
• Severe liver dysfunction
• Active malignancy
• Pregnancy (safe to consider pregnancy >10 days after ceasing treatment)
• Macula oedema
o ?previous uveitis
Interactions:
• Class Ia (e.g. quinidine, procainamide),
• Class III anti-arrhythmic drugs (e.g. amiodarone, sotalol),
• QT prolonging drugs with known arrhythmogenic properties, heart rate lowering calcium channel blockers (such as verapamil or diltiazem) or other substances which may decrease heart rate (e.g. ivabradine or digoxin).
• Beta blockers – can be started with caution once patient stable on siponimod
• CYP2C9 and CYP3A4 inducers
o Generally not a problem – Some (e.g. Carbamazepine and modafinil) may reduce siponimod efficacy
• No effect on OCP
Adverse events from EXPAND trial:
• Liver issues (12% vs 4%)
• Infections – Overall no difference from placebo
o Herpes Zoster (2% vs 1%)
• Skin cancer – no difference from placebo (1% both groups)
• Lymphopaenia – no difference from placebo (1% both groups)
• Macular Oedema – 2% vs 1%
• Peripheral oedema – 5% vs 2%
• Hypertension – 12% vs 9%
• Arrhythmia
o Bradycardia during treatment initiation – 4% vs 3%
o Any bradyarrhythmia – 3% vs 0.4%
PML
None yet recorded (as of Jan 2021)
• Cost: $29,000/yr (Same as fingolimod)
Pre-treatment screening
|
Test |
Reason |
|
BHCG or
urine pregnancy test |
Increased
risk of foetal malformation |
|
FBC |
Lymphopenia often occurs |
|
LFT |
Raised LFTs
can occur |
|
Hepatitis B and C |
Risk unknown, excluded from trials. |
|
VZV IgG |
Disseminated
zoster has occurred. Vaccinate if
negative. |
|
HIV |
Risk unknown, excluded from trials. |
|
Quantiferon gold |
No known
increased risk. |
|
CXR (optional) |
Any evidence of TB or other infectious disease.
Baseline. |
|
ECG |
Bradycardia
or heart block, QTc |
|
Skin Check |
Possible increased risk of skin cancer. |
|
Ophthalmology
review or macula OCT |
Baseline for
macula oedema. |
|
Respiratory function tests (optional) |
Optional, there may be slight decrease in lung
function. |
|
CYP2C9
Genotype |
*3*3 –
should not receive Rx *1*3 or *2*3
– reduced, 1mg dose *1*1, *1*2,
*2*2 – normal dose |
|
First dose Cardiac monitoring |
Required for: - Sinus
bradycardia <55bpm - First degree
heart block - Mobitz type I
heart block - Hx of heart disease >6 months ago |
Advice to patients
• Drug must be taken continuously – breaks in treatment may lead to bradycardia on restarting
• Watch for:
o Rash (particularly shingles)
o Jaundice
o Visual distortion (consider screening with Amsler grid)
• Vaccinations:
o Try and get vaccinations done prior to treatment
o Annual influenza vaccine encouraged
o Avoid live attenuated vaccines while on treatment
• Malignancy – ensure standard malignancy screening, including yearly skin check
• Women – avoid pregnancy, ensure adequate contraception, notify treating team immediately if pregnant
Teriflunomide (Aubagio, Teriflagio)
Mechanism
• Inhibits pyrimidine synthesis
Efficacy - Trials
• TEMSO trial
o 7mg or 14mg Vs Placebo (all figures below are for the 14mg dose)
o RRR ~31%
o Annual Relapse Rate from 0.5 vs 0.37
o Decrease in EDSS progression at early time points
o MRI T2 lesion accumulation over 2 years - ~64% relative reduction
• TOWER
o Teriflunomide 7mg vs 14mg vs Placebo (Results for 14mg below)
o 1169 patients
o RRR 36% (0.5 vs 0.32)
o Time to sustained disability progression – (15 vs 21%) reduced HR 0.65
o The Lancet Jan 2014
• TENERE trial
o Teriflunomide 7mg or 14mg vs IFNB-1a (Rebif) (results for 14mg shown below)
o 324 patients
o Primary outcome was “Treatment failure” (either a relapse or discontinuation) – there was no significant difference.
- With teriflunomide – less patients discontinued, however more patients relapsed
- Drop out 13.5% vs 24% (i.e. if you could stay on IFN would probably be better off?)
o Relapse rate (after adjustment for dropout) was 0.22 IFN vs 0.26 Teriflunomide
• IFN-B combined with teriflunomide vs IFN
o No major safety concerns
o Seemed to be additive effect
o Published in neurology (Freedman 2012)
Adverse events
Contraindications
• Pregnancy
• Liver dysfunction
Serious adverse effects
• Hepatotoxicity
o Mild elevations are common (although less common than IFN)
o Severe elevations occurred in ~2% and this was similar with placebo and IFN
o ?Cease if >3xULN
• Cytopaenia’s
o Neutrophils – a minor drop is common (e.g 17% to <1.5 in TOWER, average drop in TEMSO was~1.0), however severe neutropaenia only occurred in 2 patients in all trials <1%.
o Lymphocytes – a minor drop is common (avg. drop 0.3), severe lyphopaenia was not observes
Potential adverse effects:
• Respiratory disease
o Has not occurred with Teriflunomide but has occurred in Lefunomide (in patients with RA on methotrexate)
• Peripheral neuropathy/paraesthesia
• Serious Infections – there was no significant difference
• Nasopharyngitis – overall there was no significant increase across the trials
Less serious side effects
• Diarrhoea (TEMSO -Placebo vs drug; 9vs18%)
• Nausea (7% vs 14%)
• Hair thinning (3% vs 13%) (reversible)
Pregnancy
• Category X
• Has caused defects in animals
• Is excreted in semen and therefore should not be used by men wishing to farther a child.
• Not safe with breastfeeding
Treatment of toxicity
• Teriflunomide has a long half-life which is contributed to by enterohepatic cycling – can require 3months to clear
• Administer cholestyramine, 8g every 8 hours, to help wash out
Pharmacology and interactions
• Long half life ~19 days – takes 3 months to achieve steady state
• Takes average 8 months for drug to washout to a level safe for pregnancy.
• Minimal significant drug interactions
Dosing
• 14mg tablet daily
Pre-treatment screening
• MRI brain
• FBC, Electrolytes, LFT
• HBV serology
• HCV serology
• Varicella IgG
• HIV
• Quantiferon
• Pregnancy test
Ensure all appropriate vaccinations have been given
Monitoring
• FBC and LFTs every month for 6 months then every 2 months ongoing.
Dimethyl Fumarate (Tecfidera)
· Previously known as BG-12
Mechanism
• Alters cellular transcription - NFkB inhibitor, via Nrf2 stimulation
• Alters pathways involved in immune system homeostasis
• Exact mechanisms not known
Efficacy -Trials
• DEFINE
o 240 Tds vs 240 bd vs Placebo
o RRR ~50% (annual relapse rate) at 2 years
o EDSS progression reduction from 60 weeks
o MRI T2 ~85% reduction
• CONFIRM study
o Annualised RR reduction BG12 vs Glatiramer - 44% vs 29%
Adverse effects
• Flushing (5% vs 38%)
• GI symptoms ~20%
o Nausea, upper abdominal pain, diarrhoea
• The above side effects tend to decrease over the first couple of months to a rate of ~5%
• Reduced lymphocyte count ~5% of patients had count <0.5
• Minor increases in LFTs
Pharmacology and interactions
• Short half life ~1 hour
• Elimination ~60% via exhalation
Dosing
• 120mg bd as titration for 1-2 weeks
• 240mg bd ongoing
Pre-treatment screening
• MRI brain
• FBC, Electrolytes, LFT
• HBV serology
• HCV serology
• Varicella IgG
• HIV
• Quantiferon
• Consider pregnancy test
• Ensure all appropriate vaccinations have been given
Monitoring
• FBC and LFTs 6 weeks after treatment and then every 6 months
• Consider pausing treatment if lymphocyte consistently below 0.5
Advice to patients
• Consider taking an aspirin with dose if side effects are problematic
• Vaccinations:
o Try and get vaccinations done prior to treatment
o Annual influenza vaccine encouraged
o Avoid live attenuated vaccines while on treatment
• Women considering pregnancy should discuss with treating team first.
Natalizumab (Tysabri)
Mechanism
• Antibody against alpha-4 integrins – cell membrane proteins on leukocytes
• Alpha 4 integrin binds to endothelial VCAM-1 allowing ingress into the CNS
• Natalizumab thus Inhibits T and B cell entry into CNS
Efficacy - Trials
AFFIRM
• Natalizumab vs Placebo
• Annualised relapse rate 0.23 vs 0.73 as monotherapy, RRR 68%, ARR 50%
• New MRI T2 lesions at one year – 1.2 vs 6.1
• Disability progression 17% vs 29% at 2 years, RRR 42%, ARR 12%
SENTINEL study (NEJM 2006)
• Enrolled patients who had relapses despite being on interferon-Beta 1a (Avonex)
• Natalizumab vs continued use of Avonex
• ARRate 0.34 vs 0.75 (ARR 41%)
• 2 year cumulative sustained disability progression – 23 vs 29% (ARR 6%)
TOP (registry study)
Adverse effects and contraindications
• Infusion reactions – headache, nausea, dizziness, itch rash etc. (24% vs 18% in placebo)
o <1% chance of serious infusion reaction/hypersensitivity
• Possible small increased risk of other infections
• Pregnancy
o No observed increase in risk of congenital malformations
o Possible alteration in foetal blood counts – particularly if used in late pregnancy
• Breast feeding – likely to be safe in most instances, caution with premature infants.
• Risk of PML (Progressive multifocal leukoencephalopathy) – see below
PML risk stratification
• Latest data from:
Ho PR, et al. I. Risk of natalizumab-associated progressive multifocal leukoencephalopathy in patients with multiple sclerosis: a retrospective analysis of data from four clinical studies.
Lancet Neurol. 2017 Nov;16(11):925-933
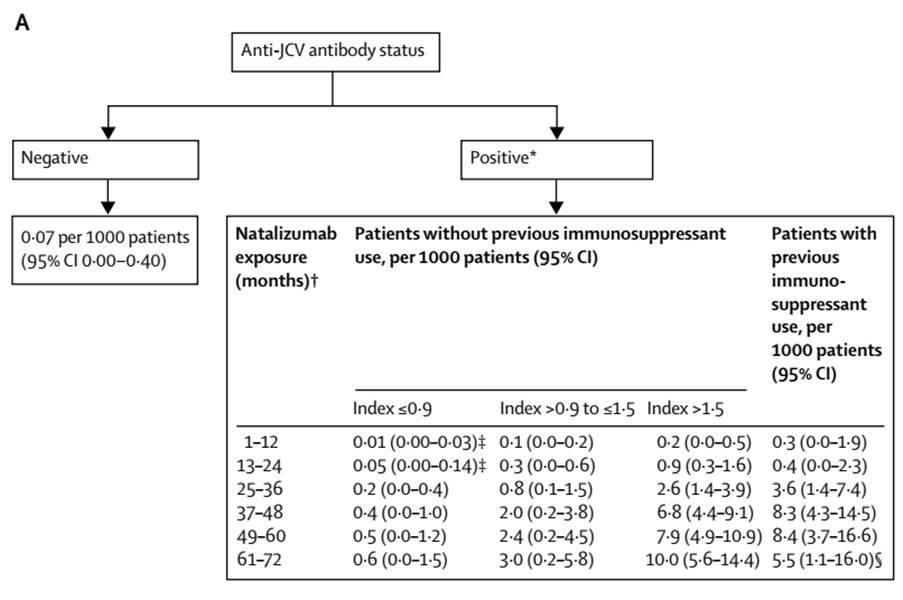
From table above – chance of PML is 1 in……
|
Time |
Index
<0.9 |
>0.9-<1.5 |
>1.5 |
Prior
immune. |
|
1-12
months |
100,000 |
10,000 |
5000 |
3333 |
|
13-24
months |
20,000 |
3333 |
1111 |
2500 |
|
25-36
months |
5000 |
1250 |
385 |
278 |
|
37-14
months |
2500 |
500 |
147 |
120 |
Pharmacology and Interactions
• Antibody with half life ~26 days
• 6% of patients develop neutralizing antibodies
Dosing
• 300mg IV every 4 weeks
Pre-treatment screening
• MRI brain (ideally with gadolinium)
• FBC, Electrolytes, LFT
• HBV serology
• HCV serology
• Varicella IgG
• HIV
• Quantiferon
• Pregnancy test or discussion
• Ensure all appropriate vaccinations have been given
Monitoring
• FBC/LFT 6 weeks after first dose, then every 6 months
o Altered blood count is common – increased lymphocytes, monocytes, eosinophils and basophils with occasional nucleated erythrocytes.
Other issues
· Rebound increase in relapse rate can occur from ~8-12 weeks – consider starting an alternative therapy early to help prevent this issue.
· Presence of a relapse during the ‘gap’ has been found to be risk factor for ongoing relapse (once starting fingolimod) (Neurology 82 1204-1211 (2014))
· If breakthrough disease occurs on natalizumab – consider testing neutralising antibodies
Advice to patients
• Vaccinations:
o Try and get vaccinations done prior to treatment
o Annual influenza vaccine encouraged
o Avoid live attenuated vaccines while on treatment
• Women considering pregnancy should discuss with treating team first.
• If abnormal blood count is detected this may be an effect of the natalizumab and should be discussed with treating team.
Alemtuzumab (Lemtrada)
Mechanism
• Anti-CD52 monoclonal antibody
• Depletes lymphocytes
o Lowest levels reached at 1 month point
o After treatment B-cells recover in 7 months, CD8 cells in 20months and CD4 in 35months.
• “After lymphocyte depletion, a distinctive pattern of T- and B- lymphocyte repopulation occurs over time, changing the balance of the immune system”
Efficacy Trials
• Proven efficacy in RCTs vs interferon
• ARR % reduction ranging from 50-69%
• Reduction in MRI lesions
• Indication of reduction in disability progression
Major Phase 2/3 trials
· All vs Rebif (IFN-1a S/C)
|
|
CAMMS223 |
CAMMS223
-extension |
CARE-MS
I |
CARE-MS
II |
||||
|
|
3
years |
5
years |
2
years |
2
years |
||||
|
|
Active RRMS (2relapses in 2 yrs, at least 1 Gad+
lesion), Rx naïve, EDSS </= 3, Onset </=3yrs |
Active RRMS (2relapses in prior 2 yrs, 1 relapse in
last yr), Rx naïve, EDSS </=3,
Onset </= 5yrs |
Active RRMS (same as CARE-MSI), relapsing on prior DMT, EDSS </=
5, Onset </= 10yrs |
|||||
|
Rx (Patient
numbers) |
IFN
(111) |
AL
(112) |
IFN (111) |
AL (112) |
IFN (187) |
AL (376) |
IFN (202) |
AL
(426) |
|
ARR |
0.36 |
0.11 |
0.35 |
0.12 |
0.39 |
0.18 |
0.52 |
0.26 |
|
ARR
% reduction |
|
69 |
|
66 |
|
55 |
|
49 |
|
ARR
% absolute |
|
25 |
|
23 |
|
21 |
|
26 |
|
Relapse
free % |
52 |
77 |
41 |
68 |
|
(NS) |
|
42 |
|
SAD% |
24 |
8 |
30 |
13 |
11 |
8 |
21 |
13 |
|
SAD
RRR% |
|
67 |
|
57 |
|
30
(NS) |
|
42 |
|
EDSS
score change |
+0.46 |
-0.32 |
+0.46 |
-0.15 |
NS |
NS |
+0.24 |
-0.17 |
|
MSFC
change |
- |
- |
- |
- |
+0.03 |
+0.11 |
-0.04 |
+0.08 |
|
SRD% |
- |
- |
- |
- |
25 |
23
|
13 |
29 |
|
|
|
|
|
|
|
|
|
|
|
New
or enlarging T2 lesions % |
|
|
|
|
58 |
49 |
68 |
46 |
|
T2
lesion volume median change % |
|
|
|
|
-6.5 |
-9.3 |
-1.2 |
-1.3 |
|
Median
change in brain parenchyma% |
|
|
|
|
-1.5 |
-0.9 |
-0.8 |
-0.6 |
|
|
|
|
|
|
|
|
|
|
|
MRI
and clinically disease free % |
|
|
|
|
27 |
39 |
14 |
32 |
|
Hospitalisation
reduction |
|
|
|
|
|
29
(NS) |
|
55 |
|
|
|
|
|
|
|
|
|
|
SAD = sustained accumulation of disability, SRD = sustained reduction in disability
Adverse effects and contraindications
Contraindications
• Hypersensitivity to alemtuzumab
• HIV infection
• Relative CI:
o Active autoimmune disease
o Active or latent infection
o Anti-TPO antibodies
o Platelet count <100,000
o Poorly controlled asthma
Adverse effects
•
Infusion associated reactions
o Cytokine release due to lymphocyte death and nitric oxide release
o Most patients will have some degree of reaction, significant in ~20%
o Can cause transient worsening of pre-existing symptoms
o Reduce with subsequent infusions
o (See below for specific management)
•
Arterial dissection and stroke
o Rare cases of stroke occurring within 1 week (usually 3 days) of infusion
o Often associated with cervicocephalic arterial dissection.
•
Autoimmunity complications
o Autoimmune thyroid disease
- ~ 36%
- Peak at 3 years
o ITP
- 1.7%
- First case died as was unrecognised
o Nephropathy
- 7 cases (by 7/2015) ~ Incidence ~0.3%
- Anti-GBM
- Membranous Golmerulonerphritis
- 2 early cases required renal transplant – not identified early
•
Infection
o Increased rate of mild to moderate infections (77% vs 66% in IFN group)
o Serious infections ~2.7% (vs 1%)
- Peak at 4 weeks post infusion
o Herpes zoster ~0.2-0.3%/year
- Give oral prophylaxis
o Pneumonia
o TB (one case)
o Listeria
o Superficial fungal infections 12% (vs 3%) – e.g. candidiasis
o Slight reduction as time goes by
• Bradycardia
o Has been identified in follow-up studies
o Never been symptomatic
• Cancer
o No overall increased risk of cancer
o Increased risk BCC - slight
o Increased thyroid cancers – probably because of increased surveillance
o HPV associated cervical dysplasia – suggest annual cervical smear
• Other
o Pneumonitis ?association
o Haemophagocytic lymphohistiocytosis
o Acute acalculous chlocystitis (0.2%)
Vaccination
• Able to mount good immune response when challenged – therefore vacccinations can still be given
Pregnancy
· Women should delay becoming pregnant until 4 months after treatment
Pharmacology and Interactions
• Antibody with rapid clearance – essentially eliminated by 30 days.
Dosing and Monitoring
Pre-initiation
|
VZV
serology |
VZV
neg - Vaccinate prior to Rx |
|
HBV,
HCV, HIV serology |
|
|
Quantiferon (TB test) |
|
|
HPV
screening (Pap smear) |
|
|
FBC |
|
|
Electrolytes |
|
|
Urinalysis
with microscopy |
|
|
TFTs
(including anti-TPO antibodies) |
|
|
CXR |
|
|
Pregnancy test |
|
• Complete vaccinations 6 weeks prior to dose, consider:
o VZV
o HBV/HAV
• Women – should use effective contraception during infusion and for 4 months post treatment
Infusion
Dosing schedule
• 12mg/day for 5 days (days 1-3 q4h, day 4 q3h, day 5 q2h)
• 12/mg/day for 3 days at 12 months
• Optional: 12mg/day for 3 days at 24 months.
During treatment
• Premedication
o Methylprednisolone 1g IV over 1 hour in 100ml N/saline prior to first 3 infusions
o Daily antihistamine loratadine 10mg (+/- ranitidine 300mg)
o Paracetamol 1g PO + PRN
o Anti-viral prophylaxis – acyclovir 200mg bd (or valacyclovir 500mg bd) – for 1 month
o Encourage fluids – 3L/24hours
• Observations
o 30min observations during infusion and one hour after
• Treatment of specific side effects:
o Pyrexia
- Low grade – paracetamol
- Persistent high grade – stop infusion
o Rash – additional antihistamines
o Headache – paracetamol
o Nausea – antiemetic
o Insomnia – temazepam
o Chest tightness - salbutamol nebs if wheezy
o Thrombophebitis – may need to change canula site.
o Hypotension
- Stop infusion
- IV fluids (500mls stat + PRN)
o Marked respiratory symptoms, oxygen desaturation
-
stop infusion
- 100% oxygen
- Hydrocortisone 100mg IV + antihistamine
- Consider adrenaline 1:1000 IM if severe
o Blood tests
- transient elevation in LFTS may be seen – no action in <5xULN
- transient neutrophilia – due to steroids
- Lymphocyte count will fall.
On-going monitoring
• Monitoring required for 4 years post last infusion
|
|
Watch for: |
|
|
FBC |
|
Monthly |
|
Creatinine |
|
|
|
Monthly urinalysis and
microscopy |
|
|
|
Thyroid function (TSH) |
|
Every 3 months |
|
HPV screening |
|
Yearly |
|
|
|
|
· Absolute lymphocyte counts do NOT predict response to treatment
Other advice
• Irradiated blood products recommended - Because of the effect on the immune system it is recommended that if patients need future blood products then irradiated products are requested.
• Vaccinations - No live vaccinations for 12 months after last infusion.
• Contraception for 4 months after last infusion
Patient advice:
· Women should delay becoming pregnant until 4 months after treatment and are advised to discuss ongoing treatment plan with neurologist prior to conception.
|
(ITP)
Idiopathic Thrombocytopaenic Purpura |
Watch
For: |
Monitoring: |
|
• This is an autoimmune condition that results in a
drop in the number of platelets in the blood • Platelets are one of the components of the blood
which allow it to clot properly • Low platelets can cause bleeding |
• Bruising more easily • Bleeding from a cut that is hard to stop • Menstrual periods that are heavier, longer or more
frequent than usual • Bleeding from your gums or nose • Small scattered spots on your skin that are red,
pink or purple • Blood in your urine, stool or vomit • Coughing up blood |
Monthly
measurement of your platelet count must be done (this is part of a “Full
blood count or FBC) |
|
Nephropathies
(Kidney Disease) |
|
|
|
Antibodies
can attack the kidney and cause it to dysfunction |
• Blood in your urine |
Monthly
testing of kidney function from a blood test Monthly
testing of protein and blood in urine |
|
Thyroid
disorders |
|
|
|
Can
be either • High thyroid function • Low thyroid function |
|
Every
3 months your thyroid function should be tested on a blood test. |
|
Infection |
|
|
|
Because
of its effect on your immune cells some infections are more common and severe |
Fever Chils Muscle
aches Cough Oral
or vaginal thrush Rash Cold
sores |
Testing
can be done depending on your symptoms |
|
Blood
products |
|
|
|
Because of the effect on the immune system it
is recommended that if patients need future blood products then irradiated
products are requested. |
|
|
|
Pregnancy |
|
|
|
Women should delay becoming pregnant until 4
months after treatment and are advised to discuss ongoing treatment plan with
neurologist prior to conception |
|
No known risk of malformations. Risk of
atypical infections in pregnancy. |
Ocrelizumab (Ocrevus)
Mechanism
• Recombinant, humanised anti-CD20 monoclonal antibody (IgG1 subtype)
• Depletes CD20 B cells
Efficacy - Trials
OPERA trials
• Two trials OPERA I and II published together in one paper (NEJM 2017)
• Total of 1656 patients recruited from 2011-2013
• Compared to Rebif
• Followed for ~2 years
Results
• Very similar for both groups
• Annualised relapse rate 0.16 vs 0.29
o Relative risk reduction 46%
o Absolute risk reduction 0.14
o NNT 7
• MRI new T2 lesions
o 0.32 vs 1.41 and 0.33 vs 1.9 (OPERA I and II)
o Relative risk reduction - 77% and 83% lower
• Disability
o Disability progression 9.1% vs 13.6% (P<0.001)
o Relative risk reduction 40%
o Absolute risk reduction 4.5% (NNT 22) (Over 2 years)
• Side effects
o Infusion reactions ~30%
o No clear signal in increased in infections
Adverse effects
• Infusion reactions (pruritus, rash, throat irritation, dyspnoea, pharyngeal or laryngeal oedema, flushing, hypotension, pyrexia, fatigue, headache, dizziness, nausea and tachycardia)
o 27% with first infucion
o <10% by dose 4
• Infection
o In the trials URTI was the only consistently increased infection (~15% vs 10% - risk 5% or 1/20)
o Possible trend towards increase of other infections (including VZV)
• Possible risks based on long term use of rituximab:
o PML
o Hypogammaglobulinaemia
o Rare, severe sinopulmonary infections.
• Pregnancy – No data. Advise avoid for 6 months after last infusion.
• Breast feeding – unknown, very unlikely that any would cross from ~1 month after infusion.
Pharmacology and interactions
• It is a monoclonal antibody with a half life of ~26 days
• CD 20 cells depleted by 14 days
• 90% return to baseline by 2.5 yrs
• Renal or hepatic impairment are not expected to significantly alter its metabolism
Screening prior to treatment
• Baseline MRI
• FBC
• Electrolytes
• LFT
• Screen for infections
o TB (Quantiferon +/- CXR)
o HBV
(Including HBsAg, Anti-HBc, Anti-Hbs)
o HCV
o HIV
o VZV (vaccinate if not immune)
• Consider pregnancy test
• Consider skin check and other cancer screening if patient is at an increased risk
• Vaccinations should be completed 6 weeks prior to treatment. Influenza vaccine can be given while patient is on medication, but may not be as effective.
Dosing
• 300mg IV Infusion on day 1 and 15 and then single, 600mg infusion every 6 months
• Premedication:
o Methylprednisolone 100mg IV 30 min prior
o Anti-histamine (e.g. loratadine 10mg) 30-60min prior
o Consider paracetamol 30-60min prior
• It is likely that B-cell populations regenerate at different rates in different individuals. B-cell subset levels may indicate this, however have not be studied for ocrelizumab.
Monitoring
• Routine FBC and electrolytes ~6 weeks after initial treatment then prior to next dose
Advice for patients
o Try and get vaccinations done prior to treatment
o Annual influenza vaccine encouraged
o Avoid live attenuated vaccines while on treatment
• Ensure cancer screening is completed as per general population guidelines, annual skin check
• Ensure early investigation and treatment of respiratory or other infections. See your doctor if recurrent infections.
• Women – ensure adequate contraception for 6 months after last dose.
Ofatumumab (Kesimpta)
Efficacy
ASCLEPIOS Trials
• Ofatumumab vs teriflunomide
• 20mg days 1, 7, 14 then every 4 weeks
• Median follow-up 1.6 years
• Relapse rate:
o Relative risk reductionin two trials 50% and 60%
o Absolute risk reduction 11% and 15%
• Disability worsening confirmed at 6 months – Pooled relative risk reduction 32%, Absolute risk reduction 3.9%
• Side effects:
o Injection site reactions 20% (vs 15% placebo injection)
• Safety
o No difference in infections from teriflunomide
Screening prior to treatment
• As per recommendations for ocrelizumab (see above)
Dosing
• 20mg subcutaneous injection at week 0, 1, 2, 4 then every 4 weeks.
• Give first dose under medical supervision
Monitoring
• B-cell depletion occurs 1-2 weeks after treatment
• Median time to recovery of B-cells 24-40 weeks.
Cladribine
(Mavenclad)
Mechanism
• Nucleoside analogue
• Needs to phosphorylated to become active – requires deoxycytidine (DCK)
• In active form cladribine interferes with DNA synthesis resulting in cell death
• Lymphocytes have high DCK activity and thus are particularly susceptible
• T and B-cells are depleted and slowly repopulate over many months.
o CD4+ T-cells are depleted the most and have the slowest recovery.
Efficacy
Studied in phase 3 trial (CLARITY ~900 patients treated and then extension trial, CLARITY EXT) and demonstrated to have efficacy in relapsing remitting multiple sclerosis.
• 3.5mg/kg vs 5.25mg/kg vs placebo (~430pts per group)
• ARR 0.14 vs 0.15 vs 0.33
• Reduction in ARR 57.6% and 54.5%
• Free from 3 month sustained EDSS increase – 85.7% vs 84.9% vs 79.4%
Adverse effects
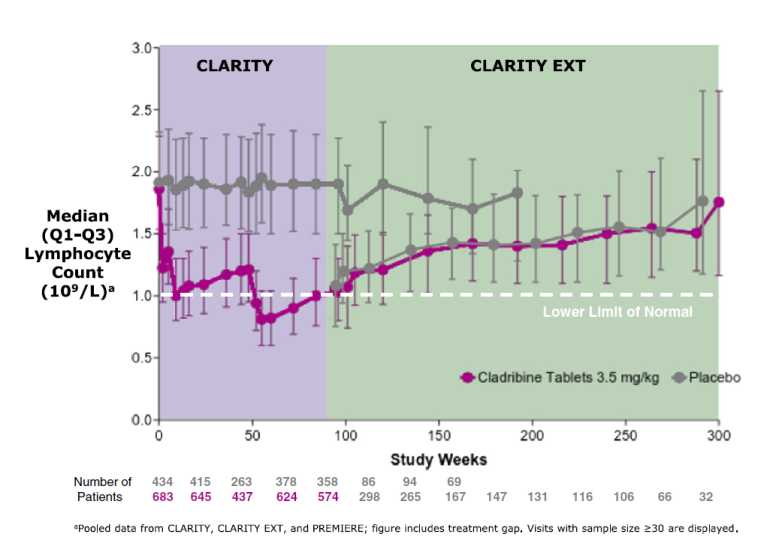
• Lymphopaenia
o Grade 3 (<0.5) in 20-25%
o Grade 4 (<0.2)in <1%
o Peak incidence 2 months after treatment
o Counts return to normal after a median 84 weeks (75% back to normal after 144 weeks)
• May be mild decrease in other blood components
• Infections
o Increased rate of herpes zoster infections
o No significant increase in other infections, however could cause difficulty managing a chronic infection if acquired during treatment course (e.g. HBV, HCV etc)
• Malignancy
o Slight excess of a malignancies in patients treated with cladribine (0.14/100patient years = 1 case for every 714 years of treatment)
Use in:
• Pregnancy – No data, however due to mechanism of action might be expected to have a teratogenic effect. Pregnancy contraindicated for 6 months after treatment.
• Breastfeeding – effect unknown.
• Male fertility – No data. Recommended avoid planning pregnancy until 6 months after treatment.
Pharmacology and interactions
• Eliminated via both renal and hepatic routes
• Suggested to avoid in moderate or severe renal impairment (GRF <60)
• Suggested to avoid in moderate or severe hepatic impairment
Interactions:
• Cladribine may be affected by following medications and combination should be avoided if possible:
• Dipyridamole, dilazep, nifedipine, cilostazol, sulindac, reserpine, eltrombopag, steroids, rifampicin, St john’s wort.
• Effect on OCP unknown – may decrease effectiveness
Screening prior to treatment
• Baseline MRI
• FBC
• Electrolytes (CI in moderate or severe renal impairment)
• LFT (CI in moderate or severe hepatic impairment)
• Screen for infections
o TB (Quantiferon +/- CXR)
o HBV
o HCV
o HIV
o VZV (vaccinate if not immune and then wait 6 weeks)
• Pregnancy test
• Consider skin check and other cancer screening if patient is at an increased risk
• Assess need for other live/live attenuated vaccines as they are contraindicated after treatment starts.
Dosing
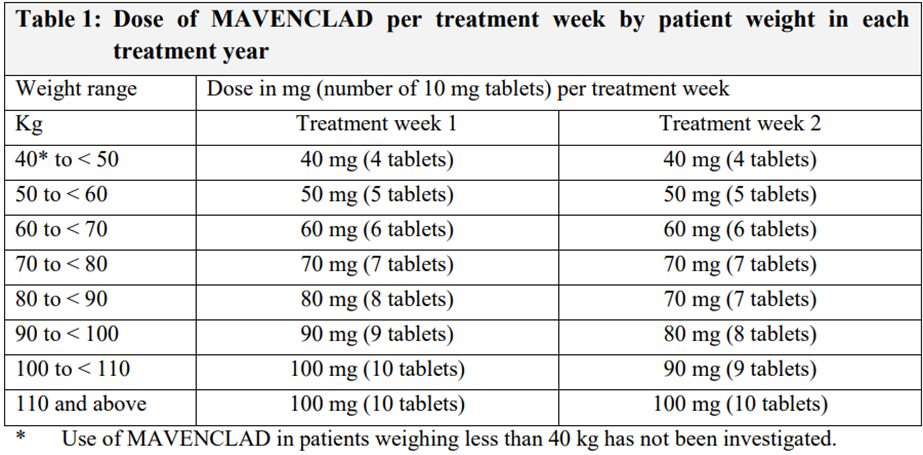
• 3.5mg/kg over 2 years (1.75mg/kg per year)
• 2 treatment courses per year (4 total treatment courses)
• 1st course over 4-5 days (week 1) followed by an identical second course 4 weeks later (week 5)
• No further treatment in years 3 and 4. Treatment beyond 4 years has not been studied (and is not currently recommended)
• Dosage is based on patient weight (see table below) – this determines the number of days (see second table)
• Can be taken with or without food
• If a days dose is missed it should be taken the next day and the course extended by a day
• Separate from any other tablets by 3 hours to avoid interactions
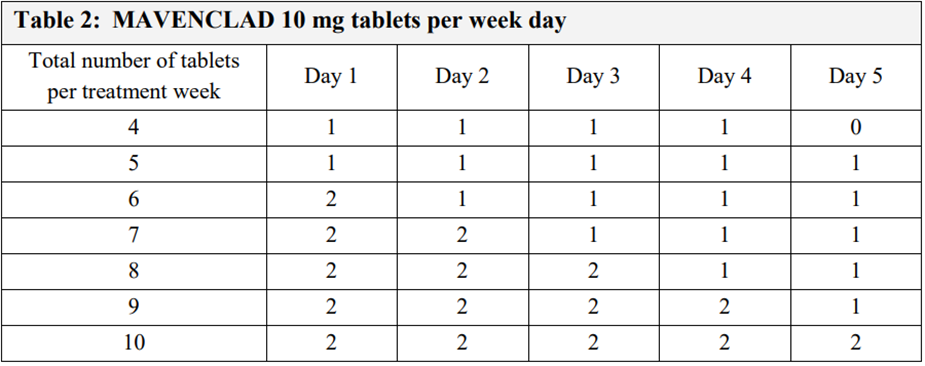
Monitoring
Malignancies
▪ Encourage usual, age appropriate cancer screening
▪ Consider yearly skin checks
Lymphocyte count
▪ Prior to initiation
▪ 2 months after treatment initiation
▪ 6 months after treatment
▪ Prior to initiation in year 2
▪ 2 months after treatment initiation
▪ 6 months after treatment
▪ More frequently if count drops to <0.5
▪ If lymphocyte count drops <0.2 consider anti-herpes prophylaxis
Advice for patients
- Monitor for signs of infection and seek early investigation and treatment
- Avoid any live or live attenuated vaccines
- Consider yearly skin checks and other appropriate cancer screening.
- Males
- Pregnancy of partner is not recommended until 6 months after treatment
- Females
- If using OCP a second form of contraception is recommended for 4 weeks after treatment as effectiveness may be decreased
- Pregnancy is not recommended until 6 months after treatment
Daclizumab (Zinbryta) - DISCONTINUED
** Discontinued due to cases of hepatic failure **
• Anti-high affinity IL2 receptor antibody
• Initially thought to reduce T cells
• ?more effect on NK cells and other lymphoid activator cells.
Trials
DECIDE Trial
• NEJM 2015
• Daclizumab vs Interferon Beta 1-a (Avonex)
• Recruited 2010-2012
• 1841 patients
• Follow-up median 2 years
Results
• ARR 0.22 vs 0.39
o Relative risk reduction - 45%
o Absolute risk reduction - 0.17 relapses/year
o NNT (For one year to prevent a relapse) = 5.9
• MRI new T2 lesions
o 9.4 vs 4.3
o Relative reduction of 54%
• No significant difference in disability
Adverse events
• Elevation in LFTs >5xULN:
o 6% vs 3% (Absolute diffference 3% - NNH 33)
• Skin reactions
o 37% vs 19% (Absolute difference 18% - NNH 5.6)
o Caused discontinuation in 5% (vs 1%)
o Rash and eczema main problems
o Serious cutaneous events 2% vs 1%
Mitoxantrone
· DNA topoisomerase
inhibitor
· Effective for both
RR and progressive
· Significant
toxicity and SE
· No longer used due to risk of secondary leukaemia
When to stop treatment
Predictors of relapse in
various studies
• Age - Some have found age is a risk for relapse - <45years, higher chance of relapse
• History of relapse – e.g. one study suggested no relapse in last 4 years predictive
• MRI GAD enhancing lesions – e.g. none in last 3 years predicts less relapse.